+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-31265 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of Csy-AcrIF5 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | complex / inhibitor / IMMUNE SYSTEM / IMMUNE SYSTEM-RNA-DNA complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationCRISPR-associated protein Csy1 / CRISPR-associated protein (Cas_Csy1) / CRISPR-associated protein Csy2 / CRISPR-associated protein (Cas_Csy2) / CRISPR-associated protein Csy3 / CRISPR-associated protein (Cas_Csy3) Similarity search - Domain/homology | |||||||||
| Biological species |  | |||||||||
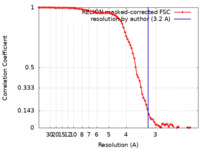
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | |||||||||
 Authors Authors | Zhang L / Feng Y | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Chem Biol / Year: 2022 Journal: Nat Chem Biol / Year: 2022Title: AcrIF5 specifically targets DNA-bound CRISPR-Cas surveillance complex for inhibition. Authors: Yongchao Xie / Laixing Zhang / Zhengyu Gao / Peipei Yin / Hao Wang / Hang Li / Zeliang Chen / Yi Zhang / Maojun Yang / Yue Feng /  Abstract: CRISPR-Cas systems are prokaryotic antiviral systems, and phages use anti-CRISPR proteins (Acrs) to inactivate these systems. Here we present structural and functional analyses of AcrIF5, exploring ...CRISPR-Cas systems are prokaryotic antiviral systems, and phages use anti-CRISPR proteins (Acrs) to inactivate these systems. Here we present structural and functional analyses of AcrIF5, exploring its unique anti-CRISPR mechanism. AcrIF5 shows binding specificity only for the target DNA-bound form of the crRNA-guided surveillance (Csy) complex, but not the apo Csy complex from the type I-F CRISPR-Cas system. We solved the structure of the Csy-dsDNA-AcrIF5 complex, revealing that the conformational changes of the Csy complex caused by dsDNA binding dictate the binding specificity for the Csy-dsDNA complex by AcrIF5. Mechanistically, five AcrIF5 molecules bind one Csy-dsDNA complex, which destabilizes the helical bundle domain of Cas8f, thus preventing subsequent Cas2/3 recruitment. AcrIF5 exists in symbiosis with AcrIF3, which blocks Cas2/3 recruitment. This attack on the recruitment event stands in contrast to the conventional mechanisms of blocking binding of target DNA. Overall, our study reveals an unprecedented mechanism of CRISPR-Cas inhibition by AcrIF5. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_31265.map.gz emd_31265.map.gz | 6.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-31265-v30.xml emd-31265-v30.xml emd-31265.xml emd-31265.xml | 18.2 KB 18.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_31265_fsc.xml emd_31265_fsc.xml | 8.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_31265.png emd_31265.png | 55 KB | ||
| Filedesc metadata |  emd-31265.cif.gz emd-31265.cif.gz | 6.5 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-31265 http://ftp.pdbj.org/pub/emdb/structures/EMD-31265 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-31265 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-31265 | HTTPS FTP |
-Validation report
| Summary document |  emd_31265_validation.pdf.gz emd_31265_validation.pdf.gz | 439.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_31265_full_validation.pdf.gz emd_31265_full_validation.pdf.gz | 439.2 KB | Display | |
| Data in XML |  emd_31265_validation.xml.gz emd_31265_validation.xml.gz | 10.5 KB | Display | |
| Data in CIF |  emd_31265_validation.cif.gz emd_31265_validation.cif.gz | 13.8 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-31265 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-31265 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-31265 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-31265 | HTTPS FTP |
-Related structure data
| Related structure data |  7eqgMC  7f45C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_31265.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_31265.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.1 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
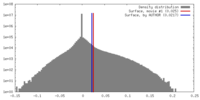
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : Csy-AcrIF5
+Supramolecule #1: Csy-AcrIF5
+Supramolecule #2: Csy-AcrIF5
+Supramolecule #3: DNA
+Macromolecule #1: CRISPR type I-F/YPEST-associated protein Csy2
+Macromolecule #2: CRISPR-associated protein Csy3
+Macromolecule #3: AcrIF5
+Macromolecule #4: type I-F CRISPR-associated endoribonuclease Cas6/Csy4
+Macromolecule #7: Type I-F CRISPR-associated protein Csy1
+Macromolecule #5: RNA (60-MER)
+Macromolecule #6: DNA (54-MER)
+Macromolecule #8: DNA (54-MER)
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 20.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: DARK FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller













 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)