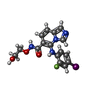
登録情報 データベース : EMDB / ID : EMD-27429タイトル Cryo-EM structure of a RAS/RAF complex (state 2) 複合体 : Kinase Complex state-1タンパク質・ペプチド : Serine/threonine-protein kinase B-rafタンパク質・ペプチド : Dual specificity mitogen-activated protein kinase kinase 1タンパク質・ペプチド : 14-3-3 protein zetaタンパク質・ペプチド : GTPase KRas isoform X2リガンド : PHOSPHOTHIOPHOSPHORIC ACID-ADENYLATE ESTERリガンド : ZINC IONリガンド : MAGNESIUM IONリガンド : 5-[(2-fluoro-4-iodophenyl)amino]-N-(2-hydroxyethoxy)imidazo[1,5-a]pyridine-6-carboxamideリガンド : PHOSPHOAMINOPHOSPHONIC ACID-GUANYLATE ESTER / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Homo sapiens (ヒト) / Spodoptera exigua (シロイチモジヨトウ)手法 / / 解像度 : 3.9 Å Eck MJ / Jeon H / Park E / Rawson S 資金援助 Organization Grant number 国 National Institutes of Health/National Cancer Institute (NIH/NCI) P50CA165962 National Institutes of Health/National Cancer Institute (NIH/NCI) R35CA242461 National Institutes of Health/National Cancer Institute (NIH/NCI) R50CA220830
ジャーナル : Nat Commun / 年 : 2023タイトル : Cryo-EM structure of a RAS/RAF recruitment complex.著者 : Eunyoung Park / Shaun Rawson / Anna Schmoker / Byeong-Won Kim / Sehee Oh / Kangkang Song / Hyesung Jeon / Michael J Eck / 要旨 : RAF-family kinases are activated by recruitment to the plasma membrane by GTP-bound RAS, whereupon they initiate signaling through the MAP kinase cascade. Prior structural studies of KRAS with RAF ... RAF-family kinases are activated by recruitment to the plasma membrane by GTP-bound RAS, whereupon they initiate signaling through the MAP kinase cascade. Prior structural studies of KRAS with RAF have focused on the isolated RAS-binding and cysteine-rich domains of RAF (RBD and CRD, respectively), which interact directly with RAS. Here we describe cryo-EM structures of a KRAS bound to intact BRAF in an autoinhibited state with MEK1 and a 14-3-3 dimer. Analysis of this KRAS/BRAF/MEK1/14-3-3 complex reveals KRAS bound to the RAS-binding domain of BRAF, captured in two orientations. Core autoinhibitory interactions in the complex are unperturbed by binding of KRAS and in vitro activation studies confirm that KRAS binding is insufficient to activate BRAF, absent membrane recruitment. These structures illustrate the separability of binding and activation of BRAF by RAS and suggest stabilization of this pre-activation intermediate as an alternative therapeutic strategy to blocking binding of KRAS. 履歴 登録 2022年6月24日 - ヘッダ(付随情報) 公開 2023年7月5日 - マップ公開 2023年7月5日 - 更新 2023年8月9日 - 現状 2023年8月9日 処理サイト : RCSB / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報
 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報 Homo sapiens (ヒト) /
Homo sapiens (ヒト) /  Spodoptera exigua (シロイチモジヨトウ)
Spodoptera exigua (シロイチモジヨトウ) データ登録者
データ登録者 米国, 3件
米国, 3件  引用
引用 ジャーナル: Nat Commun / 年: 2023
ジャーナル: Nat Commun / 年: 2023

 構造の表示
構造の表示 ダウンロードとリンク
ダウンロードとリンク emd_27429.map.gz
emd_27429.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-27429-v30.xml
emd-27429-v30.xml emd-27429.xml
emd-27429.xml EMDBヘッダ
EMDBヘッダ emd_27429.png
emd_27429.png emd_27429_additional_1.map.gz
emd_27429_additional_1.map.gz emd_27429_half_map_1.map.gz
emd_27429_half_map_1.map.gz emd_27429_half_map_2.map.gz
emd_27429_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-27429
http://ftp.pdbj.org/pub/emdb/structures/EMD-27429 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27429
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27429 emd_27429_validation.pdf.gz
emd_27429_validation.pdf.gz EMDB検証レポート
EMDB検証レポート emd_27429_full_validation.pdf.gz
emd_27429_full_validation.pdf.gz emd_27429_validation.xml.gz
emd_27429_validation.xml.gz emd_27429_validation.cif.gz
emd_27429_validation.cif.gz https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27429
https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27429 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27429
ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27429 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
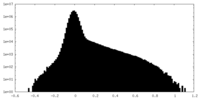
マップ ダウンロード / ファイル: emd_27429.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_27429.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) 試料の構成要素
試料の構成要素 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 画像解析
画像解析 ムービー
ムービー コントローラー
コントローラー


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)