[English] 日本語
 Yorodumi
Yorodumi- EMDB-27175: Cryo-EM structure of human Kidney Betaine-Homocysteine Methyltran... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of human Kidney Betaine-Homocysteine Methyltransferase | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | betaine-homocysteine methyltransferase / BHSMT / HYDROLASE | |||||||||
| Function / homology |  Function and homology information Function and homology information: / betaine-homocysteine S-methyltransferase / amino-acid betaine metabolic process / betaine-homocysteine S-methyltransferase activity / L-methionine salvage / amino-acid betaine catabolic process / Sulfur amino acid metabolism / Choline catabolism / 'de novo' L-methionine biosynthetic process / protein methylation ...: / betaine-homocysteine S-methyltransferase / amino-acid betaine metabolic process / betaine-homocysteine S-methyltransferase activity / L-methionine salvage / amino-acid betaine catabolic process / Sulfur amino acid metabolism / Choline catabolism / 'de novo' L-methionine biosynthetic process / protein methylation / extracellular exosome / zinc ion binding / nucleus / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
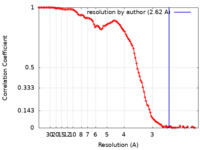
| Method | single particle reconstruction / cryo EM / Resolution: 2.62 Å | |||||||||
 Authors Authors | Lyu M / Yu EW | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Cryo-EM structure of human Kidney Betaine-Homocysteine Methyltransferase Authors: Lyu M / Yu EW | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_27175.map.gz emd_27175.map.gz | 97.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-27175-v30.xml emd-27175-v30.xml emd-27175.xml emd-27175.xml | 15.1 KB 15.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_27175_fsc.xml emd_27175_fsc.xml | 13.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_27175.png emd_27175.png | 92.8 KB | ||
| Filedesc metadata |  emd-27175.cif.gz emd-27175.cif.gz | 5.5 KB | ||
| Others |  emd_27175_half_map_1.map.gz emd_27175_half_map_1.map.gz emd_27175_half_map_2.map.gz emd_27175_half_map_2.map.gz | 95.6 MB 95.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-27175 http://ftp.pdbj.org/pub/emdb/structures/EMD-27175 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27175 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27175 | HTTPS FTP |
-Validation report
| Summary document |  emd_27175_validation.pdf.gz emd_27175_validation.pdf.gz | 745.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_27175_full_validation.pdf.gz emd_27175_full_validation.pdf.gz | 744.9 KB | Display | |
| Data in XML |  emd_27175_validation.xml.gz emd_27175_validation.xml.gz | 17.6 KB | Display | |
| Data in CIF |  emd_27175_validation.cif.gz emd_27175_validation.cif.gz | 23.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27175 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27175 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27175 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27175 | HTTPS FTP |
-Related structure data
| Related structure data |  8d45MC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_27175.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_27175.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||
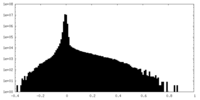
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_27175_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
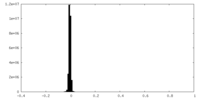
| Density Histograms |
-Half map: #1
| File | emd_27175_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
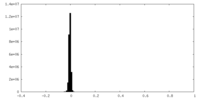
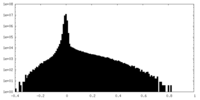
| Density Histograms |
- Sample components
Sample components
-Entire : Betaine-Homocysteine Methyltransferase
| Entire | Name: Betaine-Homocysteine Methyltransferase |
|---|---|
| Components |
|
-Supramolecule #1: Betaine-Homocysteine Methyltransferase
| Supramolecule | Name: Betaine-Homocysteine Methyltransferase / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Betaine--homocysteine S-methyltransferase 1
| Macromolecule | Name: Betaine--homocysteine S-methyltransferase 1 / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO / EC number: betaine-homocysteine S-methyltransferase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 45.059441 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MPPVGGKKAK KGILERLNAG EIVIGDGGFV FALEKRGYVK AGPWTPEAAV EHPEAVRQLH REFLRAGSNV MQTFTFYASE DKLENRGNY VLEKISGQEV NEAACDIARQ VADEGDALVA GGVSQTPSYL SCKSETEVKK VFLQQLEVFM KKNVDFLIAE Y FEHVEEAV ...String: MPPVGGKKAK KGILERLNAG EIVIGDGGFV FALEKRGYVK AGPWTPEAAV EHPEAVRQLH REFLRAGSNV MQTFTFYASE DKLENRGNY VLEKISGQEV NEAACDIARQ VADEGDALVA GGVSQTPSYL SCKSETEVKK VFLQQLEVFM KKNVDFLIAE Y FEHVEEAV WAVETLIASG KPVAATMCIG PEGDLHGVPP GECAVRLVKA GASIIGVNCH FDPTISLKTV KLMKEGLEAA RL KAHLMSQ PLAYHTPDCN KQGFIDLPEF PFGLEPRVAT RWDIQKYARE AYNLGVRYIG GCCGFEPYHI RAIAEELAPE RGF LPPASE KHGSWGSGLD MHTKPWVRAR ARKEYWENLR IASGRPYNPS MSKPDGWGVT KGTAELMQQK EATTEQQLKE LFEK QKFKS Q UniProtKB: Betaine--homocysteine S-methyltransferase 1 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 38.75 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller


 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)