[English] 日本語
 Yorodumi
Yorodumi- EMDB-24405: GltPh mutant (S279E/D405N) in complex with aspartate and sodium ions -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | GltPh mutant (S279E/D405N) in complex with aspartate and sodium ions | ||||||||||||
 Map data Map data | 20 mM HEPES/Tris pH 7.4, 250 mM NaNO3, 1 mM L-Asp Dataset A | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | outward-facing / substrate-bound / TRANSPORT PROTEIN | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationL-aspartate transmembrane transport / amino acid:sodium symporter activity / L-aspartate transmembrane transporter activity / L-aspartate import across plasma membrane / chloride transmembrane transporter activity / protein homotrimerization / chloride transmembrane transport / metal ion binding / identical protein binding / plasma membrane Similarity search - Function | ||||||||||||
| Biological species |   Pyrococcus horikoshii (archaea) / Pyrococcus horikoshii (archaea) /   Pyrococcus horikoshii (strain ATCC 700860 / DSM 12428 / JCM 9974 / NBRC 100139 / OT-3) (archaea) Pyrococcus horikoshii (strain ATCC 700860 / DSM 12428 / JCM 9974 / NBRC 100139 / OT-3) (archaea) | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.2 Å | ||||||||||||
 Authors Authors | Reddy KD / Boudker O | ||||||||||||
| Funding support |  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: J Gen Physiol / Year: 2022 Journal: J Gen Physiol / Year: 2022Title: The archaeal glutamate transporter homologue GltPh shows heterogeneous substrate binding. Authors: Krishna D Reddy / Didar Ciftci / Amanda J Scopelliti / Olga Boudker /  Abstract: Integral membrane glutamate transporters couple the concentrative substrate transport to ion gradients. There is a wealth of structural and mechanistic information about this protein family. Recent ...Integral membrane glutamate transporters couple the concentrative substrate transport to ion gradients. There is a wealth of structural and mechanistic information about this protein family. Recent studies of an archaeal homologue, GltPh, revealed transport rate heterogeneity, which is inconsistent with simple kinetic models; however, its structural and mechanistic determinants remain undefined. Here, we demonstrate that in a mutant GltPh, which exclusively populates the outward-facing state, at least two substates coexist in slow equilibrium, binding the substrate with different apparent affinities. Wild type GltPh shows similar binding properties, and modulation of the substate equilibrium correlates with transport rates. The low-affinity substate of the mutant is transient following substrate binding. Consistently, cryo-EM on samples frozen within seconds after substrate addition reveals the presence of structural classes with perturbed helical packing of the extracellular half of the transport domain in regions adjacent to the binding site. By contrast, an equilibrated structure does not show such classes. The structure at 2.2-Å resolution details a pattern of waters in the intracellular half of the domain and resolves classes with subtle differences in the substrate-binding site. We hypothesize that the rigid cytoplasmic half of the domain mediates substrate and ion recognition and coupling, whereas the extracellular labile half sets the affinity and dynamic properties. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_24405.map.gz emd_24405.map.gz | 19.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-24405-v30.xml emd-24405-v30.xml emd-24405.xml emd-24405.xml | 20.4 KB 20.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_24405.png emd_24405.png | 118.9 KB | ||
| Filedesc metadata |  emd-24405.cif.gz emd-24405.cif.gz | 5.7 KB | ||
| Others |  emd_24405_additional_1.map.gz emd_24405_additional_1.map.gz emd_24405_additional_2.map.gz emd_24405_additional_2.map.gz emd_24405_additional_3.map.gz emd_24405_additional_3.map.gz emd_24405_additional_4.map.gz emd_24405_additional_4.map.gz | 21 MB 21 MB 21 MB 21 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-24405 http://ftp.pdbj.org/pub/emdb/structures/EMD-24405 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-24405 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-24405 | HTTPS FTP |
-Validation report
| Summary document |  emd_24405_validation.pdf.gz emd_24405_validation.pdf.gz | 345.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_24405_full_validation.pdf.gz emd_24405_full_validation.pdf.gz | 345.1 KB | Display | |
| Data in XML |  emd_24405_validation.xml.gz emd_24405_validation.xml.gz | 7.8 KB | Display | |
| Data in CIF |  emd_24405_validation.cif.gz emd_24405_validation.cif.gz | 9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-24405 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-24405 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-24405 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-24405 | HTTPS FTP |
-Related structure data
| Related structure data |  7rcpMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_24405.map.gz / Format: CCP4 / Size: 343 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_24405.map.gz / Format: CCP4 / Size: 343 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 20 mM HEPES/Tris pH 7.4, 250 mM NaNO3, 1 mM L-Asp Dataset A | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.81156 Å | ||||||||||||||||||||||||||||||||||||
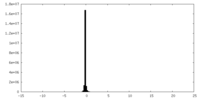
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: 20 mM HEPES/Tris pH 7.4, 250 mM NaNO3,...
| File | emd_24405_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 20 mM HEPES/Tris pH 7.4, 250 mM NaNO3, 1 mM L-Asp Dataset A, Class A2-4 | ||||||||||||
| Projections & Slices |
| ||||||||||||
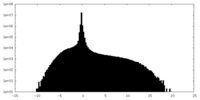
| Density Histograms |
-Additional map: 20 mM HEPES/Tris pH 7.4, 250 mM NaNO3,...
| File | emd_24405_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 20 mM HEPES/Tris pH 7.4, 250 mM NaNO3, 1 mM L-Asp Dataset A, Class A2-1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
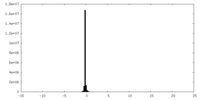
| Density Histograms |
-Additional map: 20 mM HEPES/Tris pH 7.4, 250 mM NaNO3,...
| File | emd_24405_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 20 mM HEPES/Tris pH 7.4, 250 mM NaNO3, 1 mM L-Asp Dataset A, Class A1-4 | ||||||||||||
| Projections & Slices |
| ||||||||||||
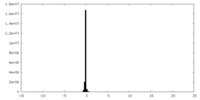
| Density Histograms |
-Additional map: 20 mM HEPES/Tris pH 7.4, 250 mM NaNO3,...
| File | emd_24405_additional_4.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 20 mM HEPES/Tris pH 7.4, 250 mM NaNO3, 1 mM L-Asp Dataset A, Class A1-8 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Complex of glutamate transporter homologue GltPh mutant (S279E/D4...
| Entire | Name: Complex of glutamate transporter homologue GltPh mutant (S279E/D405N) with sodium nitrate and aspartate |
|---|---|
| Components |
|
-Supramolecule #1: Complex of glutamate transporter homologue GltPh mutant (S279E/D4...
| Supramolecule | Name: Complex of glutamate transporter homologue GltPh mutant (S279E/D405N) with sodium nitrate and aspartate type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:   Pyrococcus horikoshii (archaea) Pyrococcus horikoshii (archaea) |
-Macromolecule #1: Glutamate transporter homolog
| Macromolecule | Name: Glutamate transporter homolog / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Pyrococcus horikoshii (strain ATCC 700860 / DSM 12428 / JCM 9974 / NBRC 100139 / OT-3) (archaea) Pyrococcus horikoshii (strain ATCC 700860 / DSM 12428 / JCM 9974 / NBRC 100139 / OT-3) (archaea)Strain: ATCC 700860 / DSM 12428 / JCM 9974 / NBRC 100139 / OT-3 |
| Molecular weight | Theoretical: 43.927055 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GLYRKYIEYP VLQKILIGLI LGAIVGLILG HYGYAHAVHT YVKPFGDLFV RLLKMLVMPI VFASLVVGAA SISPARLGRV GVKIVVYYL LTSAFAVTLG IIMARLFNPG AGIHLAVGGQ QFQPHQAPPL VHILLDIVPT NPFGALANGQ VLPTIFFAII L GIAITYLM ...String: GLYRKYIEYP VLQKILIGLI LGAIVGLILG HYGYAHAVHT YVKPFGDLFV RLLKMLVMPI VFASLVVGAA SISPARLGRV GVKIVVYYL LTSAFAVTLG IIMARLFNPG AGIHLAVGGQ QFQPHQAPPL VHILLDIVPT NPFGALANGQ VLPTIFFAII L GIAITYLM NSENEKVRKS AETLLDAING LAEAMYKIVN GVMQYAPIGV FALIAYVMAE QGVHVVGELA KVTAAVYVGL TL QILLVYF VLLKIYGIDP ISFIKHAKDA MLTAFVTRSS EGTLPVTMRV AKEMGISEGI YSFTLPLGAT INMDGTALYQ GVC TFFIAN ALGSHLTVGQ QLTIVLTAVL ASIGTAGVPG AGAIMLAMVL HSVGLPLTDP NVAAAYAMIL GIDAILDMGR TMVN VTGNL TGTAIVAKTE UniProtKB: Glutamate transporter homolog |
-Macromolecule #2: ASPARTIC ACID
| Macromolecule | Name: ASPARTIC ACID / type: ligand / ID: 2 / Number of copies: 3 / Formula: ASP |
|---|---|
| Molecular weight | Theoretical: 133.103 Da |
| Chemical component information |  ChemComp-ASP: |
-Macromolecule #3: SODIUM ION
| Macromolecule | Name: SODIUM ION / type: ligand / ID: 3 / Number of copies: 6 |
|---|---|
| Molecular weight | Theoretical: 22.99 Da |
-Macromolecule #4: water
| Macromolecule | Name: water / type: ligand / ID: 4 / Number of copies: 57 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 Component:
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sugar embedding | Material: ice | ||||||||||
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 47.91 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C3 (3 fold cyclic) / Resolution.type: BY AUTHOR / Resolution: 2.2 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 503427 |
| Initial angle assignment | Type: ANGULAR RECONSTITUTION |
| Final angle assignment | Type: ANGULAR RECONSTITUTION |
 Movie
Movie Controller
Controller




 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)