+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
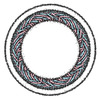
| Title | Membrane-bound CHMP2A-CHMP3 filament (430 Angstrom diameter) | ||||||||||||
 Map data Map data | Membrane-bound CHMP2A-CHMP3 filament (430 Angstrom diameter) | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | Cryo Electron Microscopy / Helical Reconstruction / Membrane-bound CHMP2A-CHMP3 filament / Negative-curvature membrane / CYTOSOLIC PROTEIN | ||||||||||||
| Function / homology | : / :  Function and homology information Function and homology information | ||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||
| Method | helical reconstruction / cryo EM / Resolution: 3.3 Å | ||||||||||||
 Authors Authors | Azad K / Desfosses A / Effantin G / Schoehn G / Weissenhorn W | ||||||||||||
| Funding support |  France, 3 items France, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2023 Journal: Nat Struct Mol Biol / Year: 2023Title: Structural basis of CHMP2A-CHMP3 ESCRT-III polymer assembly and membrane cleavage. Authors: Kimi Azad / Delphine Guilligay / Cecile Boscheron / Sourav Maity / Nicola De Franceschi / Guidenn Sulbaran / Gregory Effantin / Haiyan Wang / Jean-Philippe Kleman / Patricia Bassereau / Guy ...Authors: Kimi Azad / Delphine Guilligay / Cecile Boscheron / Sourav Maity / Nicola De Franceschi / Guidenn Sulbaran / Gregory Effantin / Haiyan Wang / Jean-Philippe Kleman / Patricia Bassereau / Guy Schoehn / Wouter H Roos / Ambroise Desfosses / Winfried Weissenhorn /   Abstract: The endosomal sorting complex required for transport (ESCRT) is a highly conserved protein machinery that drives a divers set of physiological and pathological membrane remodeling processes. However, ...The endosomal sorting complex required for transport (ESCRT) is a highly conserved protein machinery that drives a divers set of physiological and pathological membrane remodeling processes. However, the structural basis of ESCRT-III polymers stabilizing, constricting and cleaving negatively curved membranes is yet unknown. Here we present cryo-EM structures of membrane-coated CHMP2A-CHMP3 filaments from Homo sapiens of two different diameters at 3.3 and 3.6 Å resolution. The structures reveal helical filaments assembled by CHMP2A-CHMP3 heterodimers in the open ESCRT-III conformation, which generates a partially positive charged membrane interaction surface, positions short N-terminal motifs for membrane interaction and the C-terminal VPS4 target sequence toward the tube interior. Inter-filament interactions are electrostatic, which may facilitate filament sliding upon VPS4-mediated polymer remodeling. Fluorescence microscopy as well as high-speed atomic force microscopy imaging corroborate that VPS4 can constrict and cleave CHMP2A-CHMP3 membrane tubes. We therefore conclude that CHMP2A-CHMP3-VPS4 act as a minimal membrane fission machinery. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_14630.map.gz emd_14630.map.gz | 94.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-14630-v30.xml emd-14630-v30.xml emd-14630.xml emd-14630.xml | 18.7 KB 18.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_14630_fsc.xml emd_14630_fsc.xml | 27 KB | Display |  FSC data file FSC data file |
| Images |  emd_14630.png emd_14630.png | 170 KB | ||
| Filedesc metadata |  emd-14630.cif.gz emd-14630.cif.gz | 6.2 KB | ||
| Others |  emd_14630_half_map_1.map.gz emd_14630_half_map_1.map.gz emd_14630_half_map_2.map.gz emd_14630_half_map_2.map.gz | 1.4 GB 1.4 GB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-14630 http://ftp.pdbj.org/pub/emdb/structures/EMD-14630 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14630 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14630 | HTTPS FTP |
-Validation report
| Summary document |  emd_14630_validation.pdf.gz emd_14630_validation.pdf.gz | 846.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_14630_full_validation.pdf.gz emd_14630_full_validation.pdf.gz | 846.3 KB | Display | |
| Data in XML |  emd_14630_validation.xml.gz emd_14630_validation.xml.gz | 35.1 KB | Display | |
| Data in CIF |  emd_14630_validation.cif.gz emd_14630_validation.cif.gz | 47.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-14630 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-14630 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-14630 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-14630 | HTTPS FTP |
-Related structure data
| Related structure data |  7zcgMC  7zchC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_14630.map.gz / Format: CCP4 / Size: 1.7 GB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_14630.map.gz / Format: CCP4 / Size: 1.7 GB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Membrane-bound CHMP2A-CHMP3 filament (430 Angstrom diameter) | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.052 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Membrane-bound CHMP2A-CHMP3 filament (430 Angstrom diameter) half map...
| File | emd_14630_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Membrane-bound CHMP2A-CHMP3 filament (430 Angstrom diameter) half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Membrane-bound CHMP2A-CHMP3 filament (430 Angstrom diameter) half map...
| File | emd_14630_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Membrane-bound CHMP2A-CHMP3 filament (430 Angstrom diameter) half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Membrane-bound CHMP2A-CHMP3 filament (430 Angstrom Diameter) cryo...
| Entire | Name: Membrane-bound CHMP2A-CHMP3 filament (430 Angstrom Diameter) cryo-EM map |
|---|---|
| Components |
|
-Supramolecule #1: Membrane-bound CHMP2A-CHMP3 filament (430 Angstrom Diameter) cryo...
| Supramolecule | Name: Membrane-bound CHMP2A-CHMP3 filament (430 Angstrom Diameter) cryo-EM map type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Charged multivesicular body protein 2a
| Macromolecule | Name: Charged multivesicular body protein 2a / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 17.305488 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: RKTPEELLRQ NQRALNRAMR ELDRERQKLE TQEKKIIADI KKMAKQGQMD AVRIMAKDLV RTRRYVRKFV LMRANIQAVS LKIQTLKSN NSMAQAMKGV TKAMGTMNRQ LKLPQIQKIM MEFERQAEIM DMKEEMMNDA IDDAMGDE UniProtKB: UNIPROTKB: O43633 |
-Macromolecule #2: Charged multivesicular body protein 3
| Macromolecule | Name: Charged multivesicular body protein 3 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 18.429684 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: PPKELVNEWS LKIRKEMRVV DRQIRDIQRE EEKVKRSVKD AAKKGQKDVC IVLAKEMIRS RKAVSKLYAS KAHMNSVLMG MKNQLAVLR VAGSLQKSTE VMKAMQSLVK IPEIQATMRE LSKEMMKAGI IEEMLEDTFE SMDDQEEMEE EAEMEIDRIL UniProtKB: UNIPROTKB: Q9Y3E7 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
| ||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 293.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Number real images: 5028 / Average exposure time: 5.0 sec. / Average electron dose: 24.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.5 µm |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)