登録情報 データベース : EMDB / ID : EMD-26205タイトル A drug and ATP binding site in type 1 ryanodine receptor Composite map 複合体 : Ryanodine receptor 1 complex with calmodulin and calstabin-1タンパク質・ペプチド : Calmodulin-1タンパク質・ペプチド : Peptidyl-prolyl cis-trans isomerase FKBP1Aタンパク質・ペプチド : Ryanodine receptor 1リガンド : CALCIUM IONリガンド : ADENOSINE-5'-TRIPHOSPHATEリガンド : ZINC IONリガンド : CAFFEINEリガンド : 4-[(7-methoxy-2,3-dihydro-1,4-benzothiazepin-4(5H)-yl)methyl]benzoic acidリガンド : (2S)-3-(octadecanoyloxy)-2-[(9Z)-octadec-9-enoyloxy]propyl 2-(trimethylammonio)ethyl phosphate機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Oryctolagus cuniculus (ウサギ) / Homo sapiens (ヒト) / rabbit (ウサギ)手法 / / 解像度 : 2.45 Å Melville Z / Dridi H / Yuan Q / Reiken S / Anetta W / Liu Y / Clarke OB / Marks AR 資金援助 Organization Grant number 国 National Institutes of Health/National Heart, Lung, and Blood Institute (NIH/NHLBI) R01HL145473 National Institutes of Health/National Heart, Lung, and Blood Institute (NIH/NHLBI) R01DK118240 National Institutes of Health/National Heart, Lung, and Blood Institute (NIH/NHLBI) R01HL142903 National Institutes of Health/National Heart, Lung, and Blood Institute (NIH/NHLBI) R01HL140934 National Institutes of Health/National Heart, Lung, and Blood Institute (NIH/NHLBI) R01AR070194 National Institutes of Health/National Heart, Lung, and Blood Institute (NIH/NHLBI) T32 HL120826
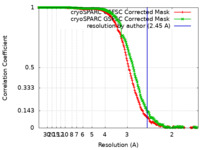
ジャーナル : Structure / 年 : 2022タイトル : A drug and ATP binding site in type 1 ryanodine receptor.著者 : Zephan Melville / Haikel Dridi / Qi Yuan / Steven Reiken / Anetta Wronska / Yang Liu / Oliver B Clarke / Andrew R Marks / 要旨 : The ryanodine receptor (RyR)/calcium release channel on the sarcoplasmic reticulum (SR) is required for excitation-contraction coupling in skeletal and cardiac muscle. Inherited mutations and stress- ... The ryanodine receptor (RyR)/calcium release channel on the sarcoplasmic reticulum (SR) is required for excitation-contraction coupling in skeletal and cardiac muscle. Inherited mutations and stress-induced post-translational modifications result in an SR Ca leak that causes skeletal myopathies, heart failure, and exercise-induced sudden death. A class of therapeutics known as Rycals prevent the RyR-mediated leak, are effective in preventing disease progression and restoring function in animal models, and are in clinical trials for patients with muscle and heart disorders. Using cryogenic-electron microscopy, we present a model of RyR1 with a 2.45-Å resolution before local refinement, revealing a binding site in the RY1&2 domain (3.10 Å local resolution), where the Rycal ARM210 binds cooperatively with ATP and stabilizes the closed state of RyR1. 履歴 登録 2022年2月15日 - ヘッダ(付随情報) 公開 2022年5月18日 - マップ公開 2022年5月18日 - 更新 2022年7月20日 - 現状 2022年7月20日 処理サイト : RCSB / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報
 マップデータ
マップデータ 試料
試料 機能・相同性情報
機能・相同性情報
 Homo sapiens (ヒト) /
Homo sapiens (ヒト) / 
 データ登録者
データ登録者 米国, 6件
米国, 6件  引用
引用 ジャーナル: Structure / 年: 2022
ジャーナル: Structure / 年: 2022
 構造の表示
構造の表示 ダウンロードとリンク
ダウンロードとリンク emd_26205.map.gz
emd_26205.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-26205-v30.xml
emd-26205-v30.xml emd-26205.xml
emd-26205.xml EMDBヘッダ
EMDBヘッダ emd_26205_fsc.xml
emd_26205_fsc.xml emd_26205_fsc_2.xml
emd_26205_fsc_2.xml FSCデータファイル
FSCデータファイル emd_26205.png
emd_26205.png emd_26205_msk_1.map
emd_26205_msk_1.map emd_26205_msk_2.map
emd_26205_msk_2.map emd_26205_msk_3.map
emd_26205_msk_3.map マスクマップ
マスクマップ emd_26205_additional_1.map.gz
emd_26205_additional_1.map.gz emd_26205_half_map_1.map.gz
emd_26205_half_map_1.map.gz emd_26205_half_map_2.map.gz
emd_26205_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-26205
http://ftp.pdbj.org/pub/emdb/structures/EMD-26205 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26205
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26205 emd_26205_validation.pdf.gz
emd_26205_validation.pdf.gz EMDB検証レポート
EMDB検証レポート emd_26205_full_validation.pdf.gz
emd_26205_full_validation.pdf.gz emd_26205_validation.xml.gz
emd_26205_validation.xml.gz emd_26205_validation.cif.gz
emd_26205_validation.cif.gz https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26205
https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26205 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26205
ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26205
 F&H 検索
F&H 検索 EMPIAR-10997 (タイトル: A drug and ATP binding site in type 1 ryanodine receptor
EMPIAR-10997 (タイトル: A drug and ATP binding site in type 1 ryanodine receptor リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_26205.map.gz / 形式: CCP4 / 大きさ: 512 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_26205.map.gz / 形式: CCP4 / 大きさ: 512 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) emd_26205_msk_1.map
emd_26205_msk_1.map emd_26205_msk_2.map
emd_26205_msk_2.map emd_26205_msk_3.map
emd_26205_msk_3.map 試料の構成要素
試料の構成要素 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 ムービー
ムービー コントローラー
コントローラー

























 Z
Z Y
Y X
X