+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
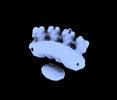
| Title | PRPH2-ROM1 octamer | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
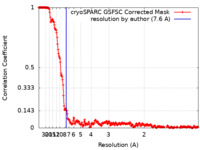
| Method | single particle reconstruction / cryo EM / Resolution: 7.6 Å | |||||||||
 Authors Authors | El Mazouni D / Gros P | |||||||||
| Funding support | European Union, 1 items
| |||||||||
 Citation Citation |  Journal: Sci Adv / Year: 2022 Journal: Sci Adv / Year: 2022Title: Cryo-EM structures of peripherin-2 and ROM1 suggest multiple roles in photoreceptor membrane morphogenesis. Authors: Dounia El Mazouni / Piet Gros /  Abstract: Mammalian peripherin-2 (PRPH2) and rod outer segment membrane protein 1 (ROM1) are retina-specific tetraspanins that partake in the constant renewal of stacked membrane discs of photoreceptor cells ...Mammalian peripherin-2 (PRPH2) and rod outer segment membrane protein 1 (ROM1) are retina-specific tetraspanins that partake in the constant renewal of stacked membrane discs of photoreceptor cells that enable vision. Here, we present single-particle cryo-electron microscopy structures of solubilized PRPH2-ROM1 heterodimers and higher-order oligomers. High-risk PRPH2 and ROM1 mutations causing blindness map to the protein-dimer interface. Cysteine bridges connect dimers forming positive-curved oligomers, whereas negative-curved oligomers were observed occasionally. Hexamers and octamers exhibit a secondary micelle that envelopes four carboxyl-terminal helices, supporting a potential role in membrane remodeling. Together, the data indicate multiple structures for PRPH2-ROM1 in creating and maintaining compartmentalization of photoreceptor cells. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_15020.map.gz emd_15020.map.gz | 615.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-15020-v30.xml emd-15020-v30.xml emd-15020.xml emd-15020.xml | 12.1 KB 12.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_15020_fsc.xml emd_15020_fsc.xml | 23.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_15020.png emd_15020.png | 35.1 KB | ||
| Others |  emd_15020_half_map_1.map.gz emd_15020_half_map_1.map.gz emd_15020_half_map_2.map.gz emd_15020_half_map_2.map.gz | 1.2 GB 1.2 GB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15020 http://ftp.pdbj.org/pub/emdb/structures/EMD-15020 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15020 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15020 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_15020.map.gz / Format: CCP4 / Size: 1.3 GB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_15020.map.gz / Format: CCP4 / Size: 1.3 GB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.656 Å | ||||||||||||||||||||||||||||||||||||
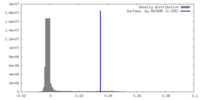
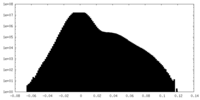
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_15020_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
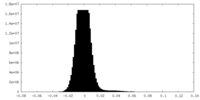
| Density Histograms |
-Half map: #2
| File | emd_15020_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
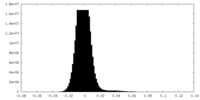
| Density Histograms |
- Sample components
Sample components
-Entire : Human PRPH2-ROM1 octamer
| Entire | Name: Human PRPH2-ROM1 octamer |
|---|---|
| Components |
|
-Supramolecule #1: Human PRPH2-ROM1 octamer
| Supramolecule | Name: Human PRPH2-ROM1 octamer / type: complex / Chimera: Yes / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.6 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Nominal defocus max: 0.8 µm / Nominal defocus min: 2.3000000000000003 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)