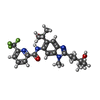+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8atn | ||||||
|---|---|---|---|---|---|---|---|
| Title | Discovery of IRAK4 Inhibitor 38 | ||||||
 Components Components | Interleukin-1 receptor-associated kinase 4 | ||||||
 Keywords Keywords | TRANSFERASE / IRAK4 / KINASE | ||||||
| Function / homology |  Function and homology information Function and homology informationMyD88 dependent cascade initiated on endosome / IRAK4 deficiency (TLR5) / TRAF6 mediated induction of NFkB and MAP kinases upon TLR7/8 or 9 activation / MyD88 cascade initiated on plasma membrane / Toll signaling pathway / interleukin-33-mediated signaling pathway / neutrophil migration / toll-like receptor 9 signaling pathway / neutrophil mediated immunity / interleukin-1 receptor binding ...MyD88 dependent cascade initiated on endosome / IRAK4 deficiency (TLR5) / TRAF6 mediated induction of NFkB and MAP kinases upon TLR7/8 or 9 activation / MyD88 cascade initiated on plasma membrane / Toll signaling pathway / interleukin-33-mediated signaling pathway / neutrophil migration / toll-like receptor 9 signaling pathway / neutrophil mediated immunity / interleukin-1 receptor binding / interleukin-1-mediated signaling pathway / MyD88-dependent toll-like receptor signaling pathway / IRAK4 deficiency (TLR2/4) / MyD88:MAL(TIRAP) cascade initiated on plasma membrane / toll-like receptor 4 signaling pathway / toll-like receptor signaling pathway / extrinsic component of plasma membrane / JNK cascade / TRAF6 mediated IRF7 activation in TLR7/8 or 9 signaling / positive regulation of smooth muscle cell proliferation / cytokine-mediated signaling pathway / Interleukin-1 signaling / PIP3 activates AKT signaling / kinase activity / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / cellular response to lipopolysaccharide / positive regulation of canonical NF-kappaB signal transduction / non-specific serine/threonine protein kinase / endosome membrane / intracellular signal transduction / innate immune response / protein serine kinase activity / protein serine/threonine kinase activity / protein kinase binding / magnesium ion binding / cell surface / extracellular space / ATP binding / nucleus / plasma membrane / cytosol / cytoplasm Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.171 Å MOLECULAR REPLACEMENT / Resolution: 2.171 Å | ||||||
 Authors Authors | Schafer, M. / Bothe, U. / Schmidt, N. / Gunther, J. / Nubbemeyer, R. / Siebeneicher, H. / Ring, S. / Boemer, U. / Peters, M. / Denner, K. ...Schafer, M. / Bothe, U. / Schmidt, N. / Gunther, J. / Nubbemeyer, R. / Siebeneicher, H. / Ring, S. / Boemer, U. / Peters, M. / Denner, K. / Himmel, H. / Sutter, A. / Terebesi, I. / Lange, M. / Wenger, A.M. / Guimond, N. / Thaler, T. / Platzek, J. / Eberspaecher, U. / Steuber, H. / Steinmeyer, A. / Zollner, T.M. | ||||||
| Funding support | 1items
| ||||||
 Citation Citation |  Journal: J.Med.Chem. / Year: 2024 Journal: J.Med.Chem. / Year: 2024Title: Discovery of IRAK4 Inhibitors BAY1834845 (Zabedosertib) and BAY1830839 . Authors: Bothe, U. / Gunther, J. / Nubbemeyer, R. / Siebeneicher, H. / Ring, S. / Bomer, U. / Peters, M. / Rausch, A. / Denner, K. / Himmel, H. / Sutter, A. / Terebesi, I. / Lange, M. / Wengner, A.M. ...Authors: Bothe, U. / Gunther, J. / Nubbemeyer, R. / Siebeneicher, H. / Ring, S. / Bomer, U. / Peters, M. / Rausch, A. / Denner, K. / Himmel, H. / Sutter, A. / Terebesi, I. / Lange, M. / Wengner, A.M. / Guimond, N. / Thaler, T. / Platzek, J. / Eberspacher, U. / Schafer, M. / Steuber, H. / Zollner, T.M. / Steinmeyer, A. / Schmidt, N. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8atn.cif.gz 8atn.cif.gz | 230.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8atn.ent.gz pdb8atn.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  8atn.json.gz 8atn.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  8atn_validation.pdf.gz 8atn_validation.pdf.gz | 1000.7 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  8atn_full_validation.pdf.gz 8atn_full_validation.pdf.gz | 1022 KB | Display | |
| Data in XML |  8atn_validation.xml.gz 8atn_validation.xml.gz | 25.4 KB | Display | |
| Data in CIF |  8atn_validation.cif.gz 8atn_validation.cif.gz | 33.4 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/at/8atn https://data.pdbj.org/pub/pdb/validation_reports/at/8atn ftp://data.pdbj.org/pub/pdb/validation_reports/at/8atn ftp://data.pdbj.org/pub/pdb/validation_reports/at/8atn | HTTPS FTP |
-Related structure data
| Related structure data |  8atbC  8atlC  8br5C  8br6C  8br7C C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 33286.582 Da / Num. of mol.: 2 / Mutation: K400A E401A E402A Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: IRAK4 / Production host: Homo sapiens (human) / Gene: IRAK4 / Production host:  References: UniProt: Q9NWZ3, non-specific serine/threonine protein kinase #2: Chemical | #3: Water | ChemComp-HOH / | Has ligand of interest | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.78 Å3/Da / Density % sol: 55.92 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop / pH: 4.9 Details: 0.1M sodium acetate buffer pH 4.9, 1.5-1.7M ammonium citrate, 0.02M hexaaminecobalt(III)chloride |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  PETRA III, EMBL c/o DESY PETRA III, EMBL c/o DESY  / Beamline: P14 (MX2) / Wavelength: 0.9763 Å / Beamline: P14 (MX2) / Wavelength: 0.9763 Å |
| Detector | Type: DECTRIS EIGER X 16M / Detector: PIXEL / Date: Oct 2, 2021 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9763 Å / Relative weight: 1 |
| Reflection | Resolution: 2.17→49.88 Å / Num. obs: 39306 / % possible obs: 99.9 % / Redundancy: 13.75 % / CC1/2: 0.999 / Rrim(I) all: 0.119 / Net I/σ(I): 13.77 |
| Reflection shell | Resolution: 2.17→2.3 Å / Num. unique obs: 6285 / CC1/2: 0.767 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: in house model Resolution: 2.171→49.875 Å / Cor.coef. Fo:Fc: 0.961 / Cor.coef. Fo:Fc free: 0.93 / SU B: 37.666 / SU ML: 0.368 / Cross valid method: FREE R-VALUE / ESU R: 0.257 / ESU R Free: 0.236 Details: Hydrogens have been added in their riding positions
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK BULK SOLVENT | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 77.038 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.171→49.875 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Total num. of bins used: 20
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller



 PDBj
PDBj