[English] 日本語
 Yorodumi
Yorodumi- PDB-7vzr: Structure of the Acidobacteria homodimeric reaction center bound ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7vzr | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structure of the Acidobacteria homodimeric reaction center bound with cytochrome c (the smaller form) | ||||||
 Components Components |
| ||||||
 Keywords Keywords | PHOTOSYNTHESIS / membrane protein | ||||||
| Function / homology |  Function and homology information Function and homology informationthylakoid / photosynthesis / electron transfer activity / iron ion binding / heme binding / membrane / metal ion binding Similarity search - Function | ||||||
| Biological species |  Chloracidobacterium thermophilum (bacteria) Chloracidobacterium thermophilum (bacteria) Chloracidobacterium thermophilum B (bacteria) Chloracidobacterium thermophilum B (bacteria) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.22 Å | ||||||
 Authors Authors | Huang, G.Q. / Dong, S.S. / Qin, X.C. / Sui, S.F. | ||||||
| Funding support | 1items
| ||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Structure of the Acidobacteria homodimeric reaction center bound with cytochrome c Authors: Dong, S. / Huang, G. / Wang, C. / Wang, J. / Sui, S.F. / Qin, X. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7vzr.cif.gz 7vzr.cif.gz | 484.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7vzr.ent.gz pdb7vzr.ent.gz | 381.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7vzr.json.gz 7vzr.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/vz/7vzr https://data.pdbj.org/pub/pdb/validation_reports/vz/7vzr ftp://data.pdbj.org/pub/pdb/validation_reports/vz/7vzr ftp://data.pdbj.org/pub/pdb/validation_reports/vz/7vzr | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  32229MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 3 types, 4 molecules AaCc
| #1: Protein | Mass: 99311.414 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Chloracidobacterium thermophilum B (bacteria) Chloracidobacterium thermophilum B (bacteria)References: UniProt: G2LDR8 #2: Protein | | Mass: 24138.053 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Chloracidobacterium thermophilum B (bacteria) Chloracidobacterium thermophilum B (bacteria)References: UniProt: G2LDR4 #7: Protein | | Mass: 15664.669 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Chloracidobacterium thermophilum B (bacteria) Chloracidobacterium thermophilum B (bacteria)References: UniProt: G2LDR3 |
|---|
-Protein/peptide , 4 types, 8 molecules EeFfGgHh
| #3: Protein/peptide | Mass: 3684.458 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Chloracidobacterium thermophilum B (bacteria) Chloracidobacterium thermophilum B (bacteria)References: UniProt: G2LK98 #4: Protein/peptide | Mass: 3885.684 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Chloracidobacterium thermophilum B (bacteria) Chloracidobacterium thermophilum B (bacteria)References: UniProt: G2LEN5 #5: Protein/peptide | Mass: 5041.152 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  Chloracidobacterium thermophilum B (bacteria) Chloracidobacterium thermophilum B (bacteria)References: UniProt: G2LJ20 #6: Protein/peptide | Mass: 1635.006 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Chloracidobacterium thermophilum (bacteria) Chloracidobacterium thermophilum (bacteria)Production host:  |
|---|
-Non-polymers , 12 types, 85 molecules 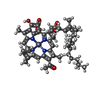



















| #8: Chemical | | #9: Chemical | ChemComp-BCL / #10: Chemical | ChemComp-CLA / #11: Chemical | #12: Chemical | #13: Chemical | ChemComp-85I / [( #14: Chemical | ChemComp-UNL / Mass: 671.023 Da / Num. of mol.: 32 / Source method: obtained synthetically / Feature type: SUBJECT OF INVESTIGATION #15: Chemical | ChemComp-85N / [( #16: Chemical | #17: Chemical | #18: Chemical | ChemComp-SF4 / | #19: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: CabRC complex / Type: COMPLEX / Entity ID: #1-#7 / Source: NATURAL |
|---|---|
| Source (natural) | Organism:  Chloracidobacterium thermophilum (bacteria) Chloracidobacterium thermophilum (bacteria) |
| Buffer solution | pH: 7.6 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2000 nm / Nominal defocus min: 500 nm / Cs: 2.7 mm |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| CTF correction | Type: NONE |
|---|---|
| 3D reconstruction | Resolution: 2.22 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 490075 / Symmetry type: POINT |
| Refinement | Highest resolution: 2.22 Å |
 Movie
Movie Controller
Controller


 PDBj
PDBj
























