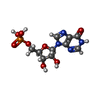
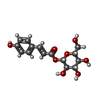
登録情報 データベース : PDB / ID : 7onfタイトル The binding of p-coumaroyl glucose to glycogen phosphorylase reveals the relationship between structural data and effects on cell metabolome Glycogen phosphorylase, muscle form キーワード 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / 生物種 Oryctolagus cuniculus (ウサギ)手法 / / / 解像度 : 1.6 Å データ登録者 Tsagkarakou, A.S. / Koulas, S.M. / Kyriakis, E. / Drakou, C.E. / Leonidas, D.D. ジャーナル : Eur J Med Chem Rep / 年 : 2021タイトル : Structure activity relationship of the binding of p-coumaroyl glucose to glycogen phosphorylase and its effect on hepatic cell metabolic pathways著者 : Tsagkarakou, A.S. / Chasapi, S.A. / Koulas, S.M. / Tsialtas, I. / Kyriakis, E. / Drakou, C.E. / Kun, S. / Somsak, L. / Spyroulias, G.A. / Psarra, A.M.G. / Leonidas, D.D. 履歴 登録 2021年5月25日 登録サイト / 処理サイト 改定 1.0 2022年4月6日 Provider / タイプ 改定 1.1 2024年1月31日 Group / Database references / Refinement descriptionカテゴリ chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / citation / pdbx_initial_refinement_model Item / _citation.journal_id_ISSN
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報 要素
要素 キーワード
キーワード 機能・相同性情報
機能・相同性情報
 X線回折 /
X線回折 /  シンクロトロン /
シンクロトロン /  分子置換 / 解像度: 1.6 Å
分子置換 / 解像度: 1.6 Å  データ登録者
データ登録者 引用
引用 ジャーナル: Eur J Med Chem Rep / 年: 2021
ジャーナル: Eur J Med Chem Rep / 年: 2021 構造の表示
構造の表示 Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク ダウンロード
ダウンロード 7onf.cif.gz
7onf.cif.gz PDBx/mmCIF形式
PDBx/mmCIF形式 pdb7onf.ent.gz
pdb7onf.ent.gz PDB形式
PDB形式 7onf.json.gz
7onf.json.gz PDBx/mmJSON形式
PDBx/mmJSON形式 その他のダウンロード
その他のダウンロード 7onf_validation.pdf.gz
7onf_validation.pdf.gz wwPDB検証レポート
wwPDB検証レポート 7onf_full_validation.pdf.gz
7onf_full_validation.pdf.gz 7onf_validation.xml.gz
7onf_validation.xml.gz 7onf_validation.cif.gz
7onf_validation.cif.gz https://data.pdbj.org/pub/pdb/validation_reports/on/7onf
https://data.pdbj.org/pub/pdb/validation_reports/on/7onf ftp://data.pdbj.org/pub/pdb/validation_reports/on/7onf
ftp://data.pdbj.org/pub/pdb/validation_reports/on/7onf
 F&H 検索
F&H 検索 リンク
リンク 集合体
集合体

 要素
要素
 X線回折 / 使用した結晶の数: 1
X線回折 / 使用した結晶の数: 1  試料調製
試料調製 シンクロトロン / サイト:
シンクロトロン / サイト:  PETRA III, EMBL c/o DESY
PETRA III, EMBL c/o DESY  / ビームライン: P13 (MX1) / 波長: 0.9763 Å
/ ビームライン: P13 (MX1) / 波長: 0.9763 Å 解析
解析 分子置換
分子置換
 ムービー
ムービー コントローラー
コントローラー



 PDBj
PDBj