+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4oyb | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal Structure Analysis of the solAC | |||||||||
 Components Components | Adenylate cyclase type 10 | |||||||||
 Keywords Keywords | LYASE | |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of cardiac muscle cell contraction / mitochondrial ATP transmembrane transport / bicarbonate binding / epithelial cilium movement involved in extracellular fluid movement / neuron projection retraction / astrocyte end-foot / central region of growth cone / glucose catabolic process / positive regulation of glycogen catabolic process / regulation of mitophagy ...negative regulation of cardiac muscle cell contraction / mitochondrial ATP transmembrane transport / bicarbonate binding / epithelial cilium movement involved in extracellular fluid movement / neuron projection retraction / astrocyte end-foot / central region of growth cone / glucose catabolic process / positive regulation of glycogen catabolic process / regulation of mitophagy / regulation of membrane repolarization / adenylate cyclase / positive regulation of oxidative stress-induced neuron intrinsic apoptotic signaling pathway / basal part of cell / cAMP biosynthetic process / adenylate cyclase activity / positive regulation of ossification / neuron projection extension / : / positive regulation of vascular associated smooth muscle cell apoptotic process / positive regulation of cardiac muscle hypertrophy / positive regulation of mitochondrial depolarization / positive regulation of reactive oxygen species biosynthetic process / negative regulation of mitochondrial membrane potential / positive regulation of ATP biosynthetic process / spermatid development / positive regulation of axon extension / Hedgehog 'off' state / negative regulation of reactive oxygen species biosynthetic process / neuron projection maintenance / positive regulation of cardiac muscle cell apoptotic process / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / apical part of cell / manganese ion binding / ATPase binding / cytoskeleton / intracellular signal transduction / cilium / neuronal cell body / dendrite / perinuclear region of cytoplasm / magnesium ion binding / mitochondrion / extracellular region / ATP binding / nucleus / plasma membrane / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / Resolution: 1.7 Å SYNCHROTRON / Resolution: 1.7 Å | |||||||||
 Authors Authors | Vinkovic, M. | |||||||||
 Citation Citation |  Journal: Chemmedchem / Year: 2014 Journal: Chemmedchem / Year: 2014Title: Crystal structure of human soluble adenylate cyclase reveals a distinct, highly flexible allosteric bicarbonate binding pocket. Authors: Saalau-Bethell, S.M. / Berdini, V. / Cleasby, A. / Congreve, M. / Coyle, J.E. / Lock, V. / Murray, C.W. / O'Brien, M.A. / Rich, S.J. / Sambrook, T. / Vinkovic, M. / Yon, J.R. / Jhoti, H. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4oyb.cif.gz 4oyb.cif.gz | 215.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4oyb.ent.gz pdb4oyb.ent.gz | 169.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4oyb.json.gz 4oyb.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/oy/4oyb https://data.pdbj.org/pub/pdb/validation_reports/oy/4oyb ftp://data.pdbj.org/pub/pdb/validation_reports/oy/4oyb ftp://data.pdbj.org/pub/pdb/validation_reports/oy/4oyb | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4oyaC  4oyiC  4oymC  4oyoC  4oypC  4oywC  4oyxC  4oyzC  4oz2C  4oz3C C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 53466.832 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: ADCY10,SAC / Production host: Homo sapiens (human) / Gene: ADCY10,SAC / Production host:  |
|---|---|
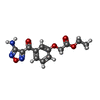
| #2: Chemical | ChemComp-GOL / |
| #3: Chemical | ChemComp-1VJ / |
| #4: Water | ChemComp-HOH / |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.64 Å3/Da / Density % sol: 53.33 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, hanging drop Details: 1ul of protein solution was mixed with 1ul of reservoir solution (0.1M sodium acetate, pH 4.8, 0.2M trisodium citrate, 16-18% PEG4K and 10% glycerol) and left to equilibrate at 4C |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID29 / Wavelength: 1.0332 Å / Beamline: ID29 / Wavelength: 1.0332 Å |
| Detector | Type: ADSC QUANTUM 4 / Detector: CCD / Date: Apr 20, 2006 |
| Radiation | Protocol: SINGLE WAVELENGTH / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.0332 Å / Relative weight: 1 |
| Reflection | Resolution: 1.7→86.53 Å / Num. obs: 60896 / % possible obs: 99.4 % / Redundancy: 2.7 % / Net I/σ(I): 12.9 |
- Processing
Processing
| Software | Name: REFMAC / Version: 5.8.0064 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 1.7→28.748 Å / Cor.coef. Fo:Fc: 0.964 / Cor.coef. Fo:Fc free: 0.946 / SU B: 4.234 / SU ML: 0.071 / Cross valid method: THROUGHOUT / ESU R: 0.098 / ESU R Free: 0.101 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: BABINET MODEL WITH MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 29.63 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: 1 / Resolution: 1.7→28.748 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj