[English] 日本語
 Yorodumi
Yorodumi- EMDB-39919: SARS-CoV-2 Omicron BA.2 spike trimer (6P) in complex with D1F6 Fa... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | SARS-CoV-2 Omicron BA.2 spike trimer (6P) in complex with D1F6 Fab, focused refinement of RBD region | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | Spike protein / Antibody Fab fragment / Complex / VIRAL PROTEIN/IMMUNE SYSTEM / VIRAL PROTEIN-IMMUNE SYSTEM complex | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationMaturation of spike protein / viral translation / Translation of Structural Proteins / Virion Assembly and Release / host cell surface / host extracellular space / suppression by virus of host tetherin activity / Induction of Cell-Cell Fusion / structural constituent of virion / entry receptor-mediated virion attachment to host cell ...Maturation of spike protein / viral translation / Translation of Structural Proteins / Virion Assembly and Release / host cell surface / host extracellular space / suppression by virus of host tetherin activity / Induction of Cell-Cell Fusion / structural constituent of virion / entry receptor-mediated virion attachment to host cell / host cell endoplasmic reticulum-Golgi intermediate compartment membrane / receptor-mediated endocytosis of virus by host cell / Attachment and Entry / membrane fusion / positive regulation of viral entry into host cell / receptor-mediated virion attachment to host cell / receptor ligand activity / symbiont-mediated suppression of host innate immune response / host cell surface receptor binding / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell / SARS-CoV-2 activates/modulates innate and adaptive immune responses / host cell plasma membrane / virion membrane / identical protein binding / membrane / plasma membrane Similarity search - Function | ||||||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.17 Å | ||||||||||||
 Authors Authors | Liu B / Gao X / Li Z / Chen Q / He J / Xiong X | ||||||||||||
| Funding support | 3 items
| ||||||||||||
 Citation Citation |  Journal: Cell Rep / Year: 2024 Journal: Cell Rep / Year: 2024Title: An unconventional VH1-2 antibody tolerates escape mutations and shows an antigenic hotspot on SARS-CoV-2 spike. Authors: Banghui Liu / Xuefeng Niu / Yijun Deng / Zhaoyong Zhang / Yanqun Wang / Xijie Gao / Huan Liang / Zimu Li / Qian Wang / Yuanyi Cheng / Qiuluan Chen / Shuangshuang Huang / Yingxian Pan / ...Authors: Banghui Liu / Xuefeng Niu / Yijun Deng / Zhaoyong Zhang / Yanqun Wang / Xijie Gao / Huan Liang / Zimu Li / Qian Wang / Yuanyi Cheng / Qiuluan Chen / Shuangshuang Huang / Yingxian Pan / Mengzhen Su / Xiancheng Lin / Chuanying Niu / Yinglin Chen / Wenyi Yang / Yudi Zhang / Qihong Yan / Jun He / Jincun Zhao / Ling Chen / Xiaoli Xiong /  Abstract: The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike (S) protein continues to evolve antigenically, impacting antibody immunity. D1F6, an affinity-matured non-stereotypic VH1-2 ...The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike (S) protein continues to evolve antigenically, impacting antibody immunity. D1F6, an affinity-matured non-stereotypic VH1-2 antibody isolated from a patient infected with the SARS-CoV-2 ancestral strain, effectively neutralizes most Omicron variants tested, including XBB.1.5. We identify that D1F6 in the immunoglobulin G (IgG) form is able to overcome the effect of most Omicron mutations through its avidity-enhanced multivalent S-trimer binding. Cryo-electron microscopy (cryo-EM) and biochemical analyses show that three simultaneous epitope mutations are generally needed to substantially disrupt the multivalent S-trimer binding by D1F6 IgG. Antigenic mutations at spike positions 346, 444, and 445, which appeared in the latest variants, have little effect on D1F6 binding individually. However, these mutations are able to act synergistically with earlier Omicron mutations to impair neutralization by affecting the interaction between D1F6 IgG and the S-trimer. These results provide insight into the mechanism by which accumulated antigenic mutations facilitate evasion of affinity-matured antibodies. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_39919.map.gz emd_39919.map.gz | 95.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-39919-v30.xml emd-39919-v30.xml emd-39919.xml emd-39919.xml | 17.1 KB 17.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_39919.png emd_39919.png | 15.4 KB | ||
| Filedesc metadata |  emd-39919.cif.gz emd-39919.cif.gz | 5.7 KB | ||
| Others |  emd_39919_half_map_1.map.gz emd_39919_half_map_1.map.gz emd_39919_half_map_2.map.gz emd_39919_half_map_2.map.gz | 95.6 MB 95.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-39919 http://ftp.pdbj.org/pub/emdb/structures/EMD-39919 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-39919 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-39919 | HTTPS FTP |
-Validation report
| Summary document |  emd_39919_validation.pdf.gz emd_39919_validation.pdf.gz | 870.4 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_39919_full_validation.pdf.gz emd_39919_full_validation.pdf.gz | 869.9 KB | Display | |
| Data in XML |  emd_39919_validation.xml.gz emd_39919_validation.xml.gz | 13 KB | Display | |
| Data in CIF |  emd_39919_validation.cif.gz emd_39919_validation.cif.gz | 15.3 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-39919 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-39919 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-39919 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-39919 | HTTPS FTP |
-Related structure data
| Related structure data |  8zc1MC  8zbyC  8zbzC  8zc0C  8zc2C  8zc3C  8zc4C  8zc5C  8zc6C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_39919.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_39919.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.32 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_39919_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
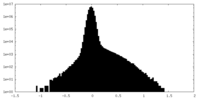
| Projections & Slices |
| ||||||||||||
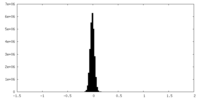
| Density Histograms |
-Half map: #2
| File | emd_39919_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
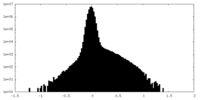
| Projections & Slices |
| ||||||||||||
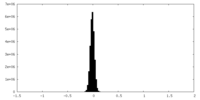
| Density Histograms |
- Sample components
Sample components
-Entire : SARS-CoV-2 Omicron BA.2 spike trimer (6P) in complex with D1F6 Fa...
| Entire | Name: SARS-CoV-2 Omicron BA.2 spike trimer (6P) in complex with D1F6 Fab, focused refinement of RBD region |
|---|---|
| Components |
|
-Supramolecule #1: SARS-CoV-2 Omicron BA.2 spike trimer (6P) in complex with D1F6 Fa...
| Supramolecule | Name: SARS-CoV-2 Omicron BA.2 spike trimer (6P) in complex with D1F6 Fab, focused refinement of RBD region type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Spike protein S1
| Macromolecule | Name: Spike protein S1 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 22.153029 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: ITNLCPFDEV FNATRFASVY AWNRKRISNC VADYSVLYNF APFFAFKCYG VSPTKLNDLC FTNVYADSFV IRGNEVSQIA PGQTGNIAD YNYKLPDDFT GCVIAWNSNK LDSKVGGNYN YLYRLFRKSN LKPFERDIST EIYQAGNKPC NGVAGFNCYF P LRSYGFRP ...String: ITNLCPFDEV FNATRFASVY AWNRKRISNC VADYSVLYNF APFFAFKCYG VSPTKLNDLC FTNVYADSFV IRGNEVSQIA PGQTGNIAD YNYKLPDDFT GCVIAWNSNK LDSKVGGNYN YLYRLFRKSN LKPFERDIST EIYQAGNKPC NGVAGFNCYF P LRSYGFRP TYGVGHQPYR VVVLSFELLH APATVCGP UniProtKB: Spike glycoprotein |
-Macromolecule #2: Light chain of D1F6 Fab
| Macromolecule | Name: Light chain of D1F6 Fab / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 23.62327 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: QPVLTQPPSA SGPPGQSVSI SCSGSRSNIG TNFVYWYQQL PGAAPKLLIY KNDQRPSGVP ERFFGSKSGT SASLAISGLR SEDEVDYYC AAWDDSLSGH VFGAGTKVTV LGTKLTVLGQ PKAAPSVTLF PPSSEELQAN KATLVCLISD FYPGAVTVAW K ADSSPVKA ...String: QPVLTQPPSA SGPPGQSVSI SCSGSRSNIG TNFVYWYQQL PGAAPKLLIY KNDQRPSGVP ERFFGSKSGT SASLAISGLR SEDEVDYYC AAWDDSLSGH VFGAGTKVTV LGTKLTVLGQ PKAAPSVTLF PPSSEELQAN KATLVCLISD FYPGAVTVAW K ADSSPVKA GVETTTPSKQ SNNKYAASSY LSLTPEQWKS HRSYSCQVTH EGSTVEKTVA PTECS |
-Macromolecule #3: Heavy chain of D1F6 Fab
| Macromolecule | Name: Heavy chain of D1F6 Fab / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 24.752727 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: EVQLVQSGAE VKKPGASVKV SCKASGYIFS DYNIHWVRQA PGQGLEWMGW ISPDSDDTNY AQSFQGRVTM TRDTSITTVY MELSSLRSD DTAVYFCARS VGYCSLNSCQ RWMWFDTWGQ GALVTVSSAS TKGPSVFPLA PSSKSTSGGT AALGCLVKDY F PEPVTVSW ...String: EVQLVQSGAE VKKPGASVKV SCKASGYIFS DYNIHWVRQA PGQGLEWMGW ISPDSDDTNY AQSFQGRVTM TRDTSITTVY MELSSLRSD DTAVYFCARS VGYCSLNSCQ RWMWFDTWGQ GALVTVSSAS TKGPSVFPLA PSSKSTSGGT AALGCLVKDY F PEPVTVSW NSGALTSGVH TFPAVLQSSG LYSLSSVVTV PSSSLGTQTY ICNVNHKPSN TKVDKKVEPK SC |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 295 K |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 4.17 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 239007 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller















 Z
Z Y
Y X
X