登録情報 データベース : EMDB / ID : EMD-35831タイトル Structure of GSK256073-GPR109A-G-protein complex 複合体 : Structure of GSK256073-GPR109A-G-protein complex複合体 : Guanine nucleotide-binding protein G(o) subunit alphaタンパク質・ペプチド : Guanine nucleotide-binding protein G(o) subunit alpha複合体 : Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1タンパク質・ペプチド : Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1複合体 : Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2タンパク質・ペプチド : Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2複合体 : ScFv16 Antibody fragmentタンパク質・ペプチド : ScFv16 Antibody Fragment複合体 : Hydroxycarboxylic acid receptor 2/GPR109Aタンパク質・ペプチド : Hydroxycarboxylic acid receptor 2リガンド : 8-chloranyl-3-pentyl-7H-purine-2,6-dione / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
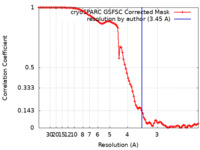
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Homo sapiens (ヒト) / Mus musculus (ハツカネズミ)手法 / / 解像度 : 3.45 Å Yadav MK / Sarma P / Chami M / Banerjee R / Shukla AK 資金援助 Organization Grant number 国 Department of Biotechnology (DBT, India) BT/PR29041/BRB/10/1697/20
ジャーナル : Nat Commun / 年 : 2024タイトル : Structure-guided engineering of biased-agonism in the human niacin receptor via single amino acid substitution.著者: Manish K Yadav / Parishmita Sarma / Jagannath Maharana / Manisankar Ganguly / Sudha Mishra / Nashrah Zaidi / Annu Dalal / Vinay Singh / Sayantan Saha / Gargi Mahajan / Saloni Sharma / Mohamed ... 著者 : Manish K Yadav / Parishmita Sarma / Jagannath Maharana / Manisankar Ganguly / Sudha Mishra / Nashrah Zaidi / Annu Dalal / Vinay Singh / Sayantan Saha / Gargi Mahajan / Saloni Sharma / Mohamed Chami / Ramanuj Banerjee / Arun K Shukla / 要旨 : The Hydroxycarboxylic acid receptor 2 (HCA2), also known as the niacin receptor or GPR109A, is a prototypical GPCR that plays a central role in the inhibition of lipolytic and atherogenic activities. ... The Hydroxycarboxylic acid receptor 2 (HCA2), also known as the niacin receptor or GPR109A, is a prototypical GPCR that plays a central role in the inhibition of lipolytic and atherogenic activities. Its activation also results in vasodilation that is linked to the side-effect of flushing associated with dyslipidemia drugs such as niacin. GPR109A continues to be a target for developing potential therapeutics in dyslipidemia with minimized flushing response. Here, we present cryo-EM structures of the GPR109A in complex with dyslipidemia drugs, niacin or acipimox, non-flushing agonists, MK6892 or GSK256073, and recently approved psoriasis drug, monomethyl fumarate (MMF). These structures elucidate the binding mechanism of agonists, molecular basis of receptor activation, and insights into biased signaling elicited by some of the agonists. The structural framework also allows us to engineer receptor mutants that exhibit G-protein signaling bias, and therefore, our study may help in structure-guided drug discovery efforts targeting this receptor. 履歴 登録 2023年4月6日 - ヘッダ(付随情報) 公開 2024年3月6日 - マップ公開 2024年3月6日 - 更新 2024年3月20日 - 現状 2024年3月20日 処理サイト : PDBj / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報
 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報 Homo sapiens (ヒト) /
Homo sapiens (ヒト) / 
 データ登録者
データ登録者 インド, 1件
インド, 1件  引用
引用 ジャーナル: Nat Commun / 年: 2024
ジャーナル: Nat Commun / 年: 2024

 構造の表示
構造の表示 ダウンロードとリンク
ダウンロードとリンク emd_35831.map.gz
emd_35831.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-35831-v30.xml
emd-35831-v30.xml emd-35831.xml
emd-35831.xml EMDBヘッダ
EMDBヘッダ emd_35831_fsc.xml
emd_35831_fsc.xml FSCデータファイル
FSCデータファイル emd_35831.png
emd_35831.png emd-35831.cif.gz
emd-35831.cif.gz emd_35831_half_map_1.map.gz
emd_35831_half_map_1.map.gz emd_35831_half_map_2.map.gz
emd_35831_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-35831
http://ftp.pdbj.org/pub/emdb/structures/EMD-35831 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35831
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35831 emd_35831_validation.pdf.gz
emd_35831_validation.pdf.gz EMDB検証レポート
EMDB検証レポート emd_35831_full_validation.pdf.gz
emd_35831_full_validation.pdf.gz emd_35831_validation.xml.gz
emd_35831_validation.xml.gz emd_35831_validation.cif.gz
emd_35831_validation.cif.gz https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35831
https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35831 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35831
ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35831 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_35831.map.gz / 形式: CCP4 / 大きさ: 91.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_35831.map.gz / 形式: CCP4 / 大きさ: 91.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) 試料の構成要素
試料の構成要素 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN ムービー
ムービー コントローラー
コントローラー



































 Z
Z Y
Y X
X