[English] 日本語
 Yorodumi
Yorodumi- EMDB-3530: Ustilago maydis kinesin-5 motor domain with N-terminal extension ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-3530 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Ustilago maydis kinesin-5 motor domain with N-terminal extension in the AMPPNP state bound to microtubules | |||||||||
 Map data Map data | Microtubule-bound Ustilago maydis kinesin-5 motor domain with N-terminal extension in the AMPPNP state | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Ustilago maydis / kinesin-5 / motor protein | |||||||||
| Function / homology |  Function and homology information Function and homology informationinitial mitotic spindle pole body separation / spindle elongation / plus-end-directed microtubule motor activity / microtubule-based movement / mitotic spindle assembly / spindle microtubule / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / mitotic spindle / structural constituent of cytoskeleton / microtubule cytoskeleton organization ...initial mitotic spindle pole body separation / spindle elongation / plus-end-directed microtubule motor activity / microtubule-based movement / mitotic spindle assembly / spindle microtubule / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / mitotic spindle / structural constituent of cytoskeleton / microtubule cytoskeleton organization / mitotic cell cycle / microtubule binding / microtubule / hydrolase activity / GTPase activity / GTP binding / ATP binding / nucleus / metal ion binding / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Ustilago maydis (fungus) / Ustilago maydis (fungus) /  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 5.1 Å | |||||||||
 Authors Authors | Moores CA / von Loeffelholz O | |||||||||
| Funding support |  United Kingdom, 1 items United Kingdom, 1 items
| |||||||||
 Citation Citation |  Journal: J Struct Biol / Year: 2019 Journal: J Struct Biol / Year: 2019Title: Cryo-EM structure of the Ustilago maydis kinesin-5 motor domain bound to microtubules. Authors: Ottilie von Loeffelholz / Carolyn Ann Moores /  Abstract: In many eukaryotes, kinesin-5 motors are essential for mitosis, and small molecules that inhibit human kinesin-5 disrupt cell division. To investigate whether fungal kinesin-5s could be targets for ...In many eukaryotes, kinesin-5 motors are essential for mitosis, and small molecules that inhibit human kinesin-5 disrupt cell division. To investigate whether fungal kinesin-5s could be targets for novel fungicides, we studied kinesin-5 from the pathogenic fungus Ustilago maydis. We used cryo-electron microscopy to determine the microtubule-bound structure of its motor domain with and without the N-terminal extension. The ATP-like conformations of the motor in the presence or absence of this N-terminus are very similar, suggesting this region is structurally disordered and does not directly influence the motor ATPase. The Ustilago maydis kinesin-5 motor domain adopts a canonical ATP-like conformation, thereby allowing the neck linker to bind along the motor domain towards the microtubule plus end. However, several insertions within this motor domain are structurally distinct. Loop2 forms a non-canonical interaction with α-tubulin, while loop8 may bridge between two adjacent protofilaments. Furthermore, loop5 - which in human kinesin-5 is involved in binding allosteric inhibitors - protrudes above the nucleotide binding site, revealing a distinct binding pocket for potential inhibitors. This work highlights fungal-specific elaborations of the kinesin-5 motor domain and provides the structural basis for future investigations of kinesins as targets for novel fungicides. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_3530.map.gz emd_3530.map.gz | 1.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-3530-v30.xml emd-3530-v30.xml emd-3530.xml emd-3530.xml | 15.6 KB 15.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_3530.png emd_3530.png | 184.9 KB | ||
| Filedesc metadata |  emd-3530.cif.gz emd-3530.cif.gz | 6.3 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-3530 http://ftp.pdbj.org/pub/emdb/structures/EMD-3530 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3530 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3530 | HTTPS FTP |
-Validation report
| Summary document |  emd_3530_validation.pdf.gz emd_3530_validation.pdf.gz | 243.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_3530_full_validation.pdf.gz emd_3530_full_validation.pdf.gz | 242.8 KB | Display | |
| Data in XML |  emd_3530_validation.xml.gz emd_3530_validation.xml.gz | 4.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-3530 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-3530 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-3530 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-3530 | HTTPS FTP |
-Related structure data
| Related structure data |  5mm7MC  3529C  5mm4C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_3530.map.gz / Format: CCP4 / Size: 1.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_3530.map.gz / Format: CCP4 / Size: 1.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Microtubule-bound Ustilago maydis kinesin-5 motor domain with N-terminal extension in the AMPPNP state | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.39 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
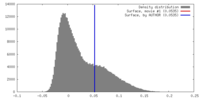
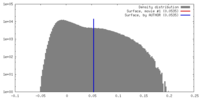
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : Ternary complex of alpha-beta-tubulin stabilized by taxol and dec...
+Supramolecule #1: Ternary complex of alpha-beta-tubulin stabilized by taxol and dec...
+Supramolecule #2: kinesin-5
+Supramolecule #3: Tubulin alpha-1A chain
+Supramolecule #4: Tubulin beta chain
+Macromolecule #1: kinesin-5
+Macromolecule #2: Tubulin alpha-1A chain
+Macromolecule #3: Tubulin beta chain
+Macromolecule #4: MAGNESIUM ION
+Macromolecule #5: PHOSPHOAMINOPHOSPHONIC ACID-ADENYLATE ESTER
+Macromolecule #6: GUANOSINE-5'-TRIPHOSPHATE
+Macromolecule #7: TAXOL
+Macromolecule #8: GUANOSINE-5'-DIPHOSPHATE
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | helical array |
- Sample preparation
Sample preparation
| Buffer | pH: 6.8 |
|---|---|
| Grid | Model: Quantifoil / Material: COPPER / Mesh: 400 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Image recording | Film or detector model: GATAN K2 QUANTUM (4k x 4k) / Average electron dose: 30.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 5.1 Å / Resolution method: FSC 0.143 CUT-OFF / Details: 13x symmetrisation / Number images used: 12629 |
| Initial angle assignment | Type: PROJECTION MATCHING |
| Final angle assignment | Type: PROJECTION MATCHING |
 Movie
Movie Controller
Controller

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)