[日本語] English
 万見
万見- EMDB-35292: Structure of C5a bound human C5aR1 in complex with Go (Composite map) -
+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Structure of C5a bound human C5aR1 in complex with Go (Composite map) | |||||||||||||||
 マップデータ マップデータ | Full map | |||||||||||||||
 試料 試料 |
| |||||||||||||||
 キーワード キーワード | GPCR / G protein / SIGNALING PROTEIN-IMMUNE SYSTEM complex | |||||||||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報complement component C5a signaling pathway / presynapse organization / regulation of tau-protein kinase activity / complement component C5a receptor activity / response to peptidoglycan / Terminal pathway of complement / membrane attack complex / sensory perception of chemical stimulus / complement receptor mediated signaling pathway / Activation of C3 and C5 ...complement component C5a signaling pathway / presynapse organization / regulation of tau-protein kinase activity / complement component C5a receptor activity / response to peptidoglycan / Terminal pathway of complement / membrane attack complex / sensory perception of chemical stimulus / complement receptor mediated signaling pathway / Activation of C3 and C5 / negative regulation of macrophage chemotaxis / vesicle docking involved in exocytosis / positive regulation of neutrophil chemotaxis / complement activation, alternative pathway / chemokine activity / G protein-coupled dopamine receptor signaling pathway / regulation of heart contraction / endopeptidase inhibitor activity / positive regulation of macrophage chemotaxis / amyloid-beta clearance / : / mu-type opioid receptor binding / corticotropin-releasing hormone receptor 1 binding / positive regulation of vascular endothelial growth factor production / negative regulation of insulin secretion / cellular defense response / G protein-coupled serotonin receptor binding / positive regulation of chemokine production / Peptide ligand-binding receptors / neutrophil chemotaxis / secretory granule membrane / locomotory behavior / complement activation, classical pathway / G protein-coupled receptor activity / Regulation of Complement cascade / muscle contraction / positive regulation of epithelial cell proliferation / astrocyte activation / microglial cell activation / G-protein beta/gamma-subunit complex binding / Olfactory Signaling Pathway / Activation of the phototransduction cascade / mRNA transcription by RNA polymerase II / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / G-protein activation / G protein-coupled acetylcholine receptor signaling pathway / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / ADP signalling through P2Y purinoceptor 12 / G beta:gamma signalling through BTK / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / Sensory perception of sweet, bitter, and umami (glutamate) taste / cognition / photoreceptor disc membrane / Adrenaline,noradrenaline inhibits insulin secretion / Glucagon-type ligand receptors / Vasopressin regulates renal water homeostasis via Aquaporins / G alpha (z) signalling events / cellular response to catecholamine stimulus / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / ADORA2B mediated anti-inflammatory cytokines production / sensory perception of taste / ADP signalling through P2Y purinoceptor 1 / adenylate cyclase-activating dopamine receptor signaling pathway / G beta:gamma signalling through PI3Kgamma / cellular response to prostaglandin E stimulus / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / GPER1 signaling / positive regulation of angiogenesis / G-protein beta-subunit binding / Inactivation, recovery and regulation of the phototransduction cascade / heterotrimeric G-protein complex / chemotaxis / G alpha (12/13) signalling events / extracellular vesicle / signaling receptor complex adaptor activity / Thrombin signalling through proteinase activated receptors (PARs) / GTPase binding / apical part of cell / retina development in camera-type eye / phospholipase C-activating G protein-coupled receptor signaling pathway / Ca2+ pathway / G alpha (i) signalling events / cell body / positive regulation of cytosolic calcium ion concentration / fibroblast proliferation / G alpha (s) signalling events / G alpha (q) signalling events / basolateral plasma membrane / killing of cells of another organism / cell population proliferation / Ras protein signal transduction / Extra-nuclear estrogen signaling / cell surface receptor signaling pathway 類似検索 - 分子機能 | |||||||||||||||
| 生物種 |  Homo sapiens (ヒト) / Homo sapiens (ヒト) /  | |||||||||||||||
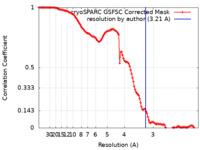
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.21 Å | |||||||||||||||
 データ登録者 データ登録者 | Yadav MK / Yadav R / Maharana J / Banerjee R / Shukla AK / Gati C | |||||||||||||||
| 資金援助 |  インド, インド,  英国, 4件 英国, 4件
| |||||||||||||||
 引用 引用 |  ジャーナル: Cell / 年: 2023 ジャーナル: Cell / 年: 2023タイトル: Molecular basis of anaphylatoxin binding, activation, and signaling bias at complement receptors. 著者: Manish K Yadav / Jagannath Maharana / Ravi Yadav / Shirsha Saha / Parishmita Sarma / Chahat Soni / Vinay Singh / Sayantan Saha / Manisankar Ganguly / Xaria X Li / Samanwita Mohapatra / Sudha ...著者: Manish K Yadav / Jagannath Maharana / Ravi Yadav / Shirsha Saha / Parishmita Sarma / Chahat Soni / Vinay Singh / Sayantan Saha / Manisankar Ganguly / Xaria X Li / Samanwita Mohapatra / Sudha Mishra / Htet A Khant / Mohamed Chami / Trent M Woodruff / Ramanuj Banerjee / Arun K Shukla / Cornelius Gati /     要旨: The complement system is a critical part of our innate immune response, and the terminal products of this cascade, anaphylatoxins C3a and C5a, exert their physiological and pathophysiological ...The complement system is a critical part of our innate immune response, and the terminal products of this cascade, anaphylatoxins C3a and C5a, exert their physiological and pathophysiological responses primarily via two GPCRs, C3aR and C5aR1. However, the molecular mechanism of ligand recognition, activation, and signaling bias of these receptors remains mostly elusive. Here, we present nine cryo-EM structures of C3aR and C5aR1 activated by their natural and synthetic agonists, which reveal distinct binding pocket topologies of complement anaphylatoxins and provide key insights into receptor activation and transducer coupling. We also uncover the structural basis of a naturally occurring mechanism to dampen the inflammatory response of C5a via proteolytic cleavage of the terminal arginine and the G-protein signaling bias elicited by a peptide agonist of C3aR identified here. In summary, our study elucidates the innerworkings of the complement anaphylatoxin receptors and should facilitate structure-guided drug discovery to target these receptors in a spectrum of disorders. | |||||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
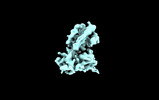
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_35292.map.gz emd_35292.map.gz | 166.9 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-35292-v30.xml emd-35292-v30.xml emd-35292.xml emd-35292.xml | 26.4 KB 26.4 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_35292_fsc.xml emd_35292_fsc.xml | 11.9 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_35292.png emd_35292.png | 37 KB | ||
| Filedesc metadata |  emd-35292.cif.gz emd-35292.cif.gz | 7.2 KB | ||
| その他 |  emd_35292_half_map_1.map.gz emd_35292_half_map_1.map.gz emd_35292_half_map_2.map.gz emd_35292_half_map_2.map.gz | 164.2 MB 164.2 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-35292 http://ftp.pdbj.org/pub/emdb/structures/EMD-35292 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35292 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35292 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_35292_validation.pdf.gz emd_35292_validation.pdf.gz | 818 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_35292_full_validation.pdf.gz emd_35292_full_validation.pdf.gz | 817.6 KB | 表示 | |
| XML形式データ |  emd_35292_validation.xml.gz emd_35292_validation.xml.gz | 20.4 KB | 表示 | |
| CIF形式データ |  emd_35292_validation.cif.gz emd_35292_validation.cif.gz | 26.7 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35292 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35292 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35292 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35292 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  8ia2MC  8i95C  8i97C  8i9aC  8i9lC  8i9sC  8j6dC  8jzzC C: 同じ文献を引用 ( M: このマップから作成された原子モデル |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_35292.map.gz / 形式: CCP4 / 大きさ: 178 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_35292.map.gz / 形式: CCP4 / 大きさ: 178 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Full map | ||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.0631 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-ハーフマップ: Half map B
| ファイル | emd_35292_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half map B | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: Half map A
| ファイル | emd_35292_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half map A | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
+全体 : C5a bound human C5aR1 in complex with Go
+超分子 #1: C5a bound human C5aR1 in complex with Go
+超分子 #2: C5a anaphylatoxin chemotactic receptor 1
+超分子 #3: C5a anaphylatoxin
+超分子 #4: Guanine nucleotide-binding protein G(o) subunit alpha
+超分子 #5: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
+超分子 #6: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
+超分子 #7: Antibody fragment - ScFv16
+分子 #1: C5a anaphylatoxin chemotactic receptor 1
+分子 #2: C5a anaphylatoxin
+分子 #3: Guanine nucleotide-binding protein G(o) subunit alpha
+分子 #4: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
+分子 #5: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
+分子 #6: Antibody fragment - ScFv16
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.4 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | TFS GLACIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: FEI FALCON IV (4k x 4k) 検出モード: COUNTING / 実像数: 10151 / 平均電子線量: 50.0 e/Å2 |
| 電子線 | 加速電圧: 200 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 3.0 µm / 最小 デフォーカス(公称値): 0.8 µm |
 ムービー
ムービー コントローラー
コントローラー







































 Z
Z Y
Y X
X