+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
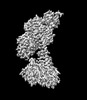
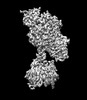
| タイトル | Focus refined map of N-lobe of human soluble guanylate cyclase in the NO+Rio state at 3.1 angstrom | ||||||||||||
 マップデータ マップデータ | |||||||||||||
 試料 試料 |
| ||||||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | ||||||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.1 Å | ||||||||||||
 データ登録者 データ登録者 | Chen L / Liu R | ||||||||||||
| 資金援助 |  中国, 3件 中国, 3件
| ||||||||||||
 引用 引用 |  ジャーナル: Nitric Oxide / 年: 2023 ジャーナル: Nitric Oxide / 年: 2023タイトル: NO binds to the distal site of haem in the fully activated soluble guanylate cyclase. 著者: Rui Liu / Yunlu Kang / Lei Chen /  要旨: Soluble guanylate cyclase (sGC) is the primary receptor for nitric oxide (NO). The binding of NO to the haem of sGC induces a large conformational change in the enzyme and activates its cyclase ...Soluble guanylate cyclase (sGC) is the primary receptor for nitric oxide (NO). The binding of NO to the haem of sGC induces a large conformational change in the enzyme and activates its cyclase activity. However, whether NO binds to the proximal site or the distal site of haem in the fully activated state remains under debate. Here, we present cryo-EM maps of sGC in the NO-activated state at high resolutions, allowing the observation of the density of NO. These cryo-EM maps show the binding of NO to the distal site of haem in the NO-activated state. | ||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_34629.map.gz emd_34629.map.gz | 59.7 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-34629-v30.xml emd-34629-v30.xml emd-34629.xml emd-34629.xml | 14.4 KB 14.4 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_34629.png emd_34629.png | 50.7 KB | ||
| マスクデータ |  emd_34629_msk_1.map emd_34629_msk_1.map | 64 MB |  マスクマップ マスクマップ | |
| その他 |  emd_34629_additional_1.map.gz emd_34629_additional_1.map.gz emd_34629_half_map_1.map.gz emd_34629_half_map_1.map.gz emd_34629_half_map_2.map.gz emd_34629_half_map_2.map.gz | 32 MB 59.5 MB 59.5 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-34629 http://ftp.pdbj.org/pub/emdb/structures/EMD-34629 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34629 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34629 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_34629_validation.pdf.gz emd_34629_validation.pdf.gz | 771 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_34629_full_validation.pdf.gz emd_34629_full_validation.pdf.gz | 770.6 KB | 表示 | |
| XML形式データ |  emd_34629_validation.xml.gz emd_34629_validation.xml.gz | 12.4 KB | 表示 | |
| CIF形式データ |  emd_34629_validation.cif.gz emd_34629_validation.cif.gz | 14.5 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34629 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34629 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34629 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34629 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_34629.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_34629.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ボクセルのサイズ | X=Y=Z: 1.045 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-マスク #1
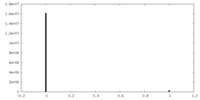
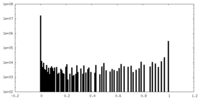
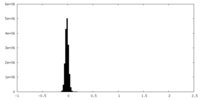
| ファイル |  emd_34629_msk_1.map emd_34629_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-追加マップ: #1
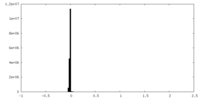
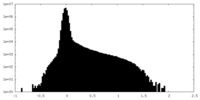
| ファイル | emd_34629_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #1
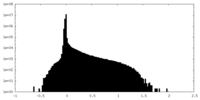
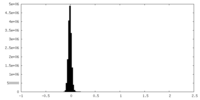
| ファイル | emd_34629_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #2
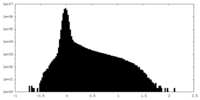
| ファイル | emd_34629_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : human soluble guanylate cyclase
| 全体 | 名称: human soluble guanylate cyclase |
|---|---|
| 要素 |
|
-超分子 #1: human soluble guanylate cyclase
| 超分子 | 名称: human soluble guanylate cyclase / タイプ: complex / ID: 1 / キメラ: Yes / 親要素: 0 / 含まれる分子: #1-#2 |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.5 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 QUANTUM (4k x 4k) 平均電子線量: 50.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 2.0 µm / 最小 デフォーカス(公称値): 1.5 µm |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 最終 再構成 | 解像度のタイプ: BY AUTHOR / 解像度: 3.1 Å / 解像度の算出法: FSC 0.143 CUT-OFF / ソフトウェア - 名称: RELION (ver. 3.0) / 使用した粒子像数: 330636 |
|---|---|
| 初期 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
| 最終 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
 ムービー
ムービー コントローラー
コントローラー
















 Z
Z Y
Y X
X