[English] 日本語
 Yorodumi
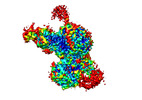
Yorodumi- EMDB-28547: Cryo-EM structure of PRC2 in complex with the long isoform of AEBP2 -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of PRC2 in complex with the long isoform of AEBP2 | ||||||||||||||||||||||||
 Map data Map data | |||||||||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||||||||
 Keywords Keywords | Polycomb repressive complex 2 Histone methyltransferase / GENE REGULATION | ||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of kidney development / hepatocyte homeostasis / cellular response to trichostatin A / regulation of gliogenesis / negative regulation of striated muscle cell differentiation / [histone H3]-lysine27 N-trimethyltransferase / sex chromatin / negative regulation of keratinocyte differentiation / random inactivation of X chromosome / CAF-1 complex ...regulation of kidney development / hepatocyte homeostasis / cellular response to trichostatin A / regulation of gliogenesis / negative regulation of striated muscle cell differentiation / [histone H3]-lysine27 N-trimethyltransferase / sex chromatin / negative regulation of keratinocyte differentiation / random inactivation of X chromosome / CAF-1 complex / histone H3K27 trimethyltransferase activity / negative regulation of retinoic acid receptor signaling pathway / regulatory ncRNA-mediated heterochromatin formation / skeletal muscle satellite cell maintenance involved in skeletal muscle regeneration / response to tetrachloromethane / cerebellar cortex development / primary miRNA binding / histone H3K27 methyltransferase activity / facultative heterochromatin formation / positive regulation of cell cycle G1/S phase transition / NURF complex / NuRD complex / regulation of cell fate specification / negative regulation of stem cell population maintenance / DNA replication-dependent chromatin assembly / ESC/E(Z) complex / chromatin silencing complex / Transcription of E2F targets under negative control by p107 (RBL1) and p130 (RBL2) in complex with HDAC1 / regulation of stem cell differentiation / RSC-type complex / protein-lysine N-methyltransferase activity / cardiac muscle hypertrophy in response to stress / negative regulation of stem cell differentiation / Transcription of E2F targets under negative control by DREAM complex / pronucleus / Polo-like kinase mediated events / synaptic transmission, GABAergic / positive regulation of dendrite development / histone H3 methyltransferase activity / lncRNA binding / negative regulation of G1/S transition of mitotic cell cycle / negative regulation of gene expression, epigenetic / G1 to G0 transition / spinal cord development / positive regulation of stem cell population maintenance / ATPase complex / G1/S-Specific Transcription / histone methyltransferase activity / Sin3-type complex / oligodendrocyte differentiation / negative regulation of transcription elongation by RNA polymerase II / Transcriptional Regulation by E2F6 / RNA Polymerase I Transcription Initiation / negative regulation of cell differentiation / histone deacetylase complex / G0 and Early G1 / subtelomeric heterochromatin formation / negative regulation of cytokine production involved in inflammatory response / RNA polymerase II core promoter sequence-specific DNA binding / pericentric heterochromatin / ribonucleoprotein complex binding / Cyclin E associated events during G1/S transition / Transcriptional regulation of brown and beige adipocyte differentiation by EBF2 / positive regulation of epithelial to mesenchymal transition / Cyclin A:Cdk2-associated events at S phase entry / keratinocyte differentiation / Deposition of new CENPA-containing nucleosomes at the centromere / enzyme activator activity / Regulation of TP53 Activity through Acetylation / protein localization to chromatin / methylated histone binding / B cell differentiation / SUMOylation of chromatin organization proteins / transcription corepressor binding / ERCC6 (CSB) and EHMT2 (G9a) positively regulate rRNA expression / positive regulation of GTPase activity / negative regulation of cell migration / PRC2 methylates histones and DNA / Regulation of PTEN gene transcription / stem cell differentiation / Defective pyroptosis / HDACs deacetylate histones / liver regeneration / promoter-specific chromatin binding / hippocampus development / transcription coregulator activity / negative regulation of transforming growth factor beta receptor signaling pathway / positive regulation of MAP kinase activity / protein modification process / positive regulation of protein serine/threonine kinase activity / brain development / regulation of circadian rhythm / heterochromatin formation / chromatin DNA binding / PKMTs methylate histone lysines / histone deacetylase binding / Activation of anterior HOX genes in hindbrain development during early embryogenesis / HCMV Early Events / cellular response to hydrogen peroxide / G1/S transition of mitotic cell cycle Similarity search - Function | ||||||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||||||||||||||
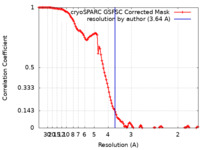
| Method | single particle reconstruction / cryo EM / Resolution: 3.64 Å | ||||||||||||||||||||||||
 Authors Authors | Boudes M / Zhang Q / Flanigan SF / Davidovich C | ||||||||||||||||||||||||
| Funding support |  Australia, 7 items Australia, 7 items
| ||||||||||||||||||||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: To be updated Authors: Boudes M / Zhang Q / Flanigan SF / Davidovich C | ||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_28547.map.gz emd_28547.map.gz | 88.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-28547-v30.xml emd-28547-v30.xml emd-28547.xml emd-28547.xml | 26.8 KB 26.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_28547_fsc.xml emd_28547_fsc.xml | 12 KB | Display |  FSC data file FSC data file |
| Images |  emd_28547.png emd_28547.png | 73.3 KB | ||
| Filedesc metadata |  emd-28547.cif.gz emd-28547.cif.gz | 8.2 KB | ||
| Others |  emd_28547_additional_1.map.gz emd_28547_additional_1.map.gz emd_28547_half_map_1.map.gz emd_28547_half_map_1.map.gz emd_28547_half_map_2.map.gz emd_28547_half_map_2.map.gz | 168.1 MB 165.1 MB 165.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-28547 http://ftp.pdbj.org/pub/emdb/structures/EMD-28547 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28547 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28547 | HTTPS FTP |
-Validation report
| Summary document |  emd_28547_validation.pdf.gz emd_28547_validation.pdf.gz | 937.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_28547_full_validation.pdf.gz emd_28547_full_validation.pdf.gz | 937.3 KB | Display | |
| Data in XML |  emd_28547_validation.xml.gz emd_28547_validation.xml.gz | 20.8 KB | Display | |
| Data in CIF |  emd_28547_validation.cif.gz emd_28547_validation.cif.gz | 26.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28547 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28547 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28547 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28547 | HTTPS FTP |
-Related structure data
| Related structure data |  8eqvMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_28547.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_28547.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.86 Å | ||||||||||||||||||||||||||||||||||||
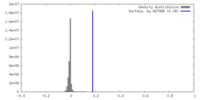
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: #1
| File | emd_28547_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
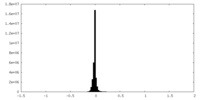
| Density Histograms |
-Half map: #2
| File | emd_28547_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
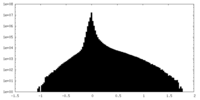
| Density Histograms |
-Half map: #1
| File | emd_28547_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
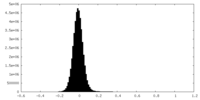
| Density Histograms |
- Sample components
Sample components
-Entire : Ternary complex of PRC2 with long isoform of AEBP2
| Entire | Name: Ternary complex of PRC2 with long isoform of AEBP2 |
|---|---|
| Components |
|
-Supramolecule #1: Ternary complex of PRC2 with long isoform of AEBP2
| Supramolecule | Name: Ternary complex of PRC2 with long isoform of AEBP2 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Histone-binding protein RBBP4
| Macromolecule | Name: Histone-binding protein RBBP4 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 47.709527 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MADKEAAFDD AVEERVINEE YKIWKKNTPF LYDLVMTHAL EWPSLTAQWL PDVTRPEGKD FSIHRLVLGT HTSDEQNHLV IASVQLPND DAQFDASHYD SEKGEFGGFG SVSGKIEIEI KINHEGEVNR ARYMPQNPCI IATKTPSSDV LVFDYTKHPS K PDPSGECN ...String: MADKEAAFDD AVEERVINEE YKIWKKNTPF LYDLVMTHAL EWPSLTAQWL PDVTRPEGKD FSIHRLVLGT HTSDEQNHLV IASVQLPND DAQFDASHYD SEKGEFGGFG SVSGKIEIEI KINHEGEVNR ARYMPQNPCI IATKTPSSDV LVFDYTKHPS K PDPSGECN PDLRLRGHQK EGYGLSWNPN LSGHLLSASD DHTICLWDIS AVPKEGKVVD AKTIFTGHTA VVEDVSWHLL HE SLFGSVA DDQKLMIWDT RSNNTSKPSH SVDAHTAEVN CLSFNPYSEF ILATGSADKT VALWDLRNLK LKLHSFESHK DEI FQVQWS PHNETILASS GTDRRLNVWD LSKIGEEQSP EDAEDGPPEL LFIHGGHTAK ISDFSWNPNE PWVICSVSED NIMQ VWQMA ENIYNDEDPE GSVDPEGQGS UniProtKB: Histone-binding protein RBBP4 |
-Macromolecule #2: Zinc finger protein AEBP2
| Macromolecule | Name: Zinc finger protein AEBP2 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 54.535496 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MAAAITDMAD LEELSRLSPL PPGSPGSAAR GRAEPPEEEE EEEEEEEEAE AEAVAALLLN GGSGGGGGGG GGGVGGGEAE TMSEPSPES ASQAGEDEDE EEDDEEEEDE SSSSGGGEEE SSAESLVGSS GGSSSDETRS LSPGAASSSS GDGDGKEGLE E PKGPRGSQ ...String: MAAAITDMAD LEELSRLSPL PPGSPGSAAR GRAEPPEEEE EEEEEEEEAE AEAVAALLLN GGSGGGGGGG GGGVGGGEAE TMSEPSPES ASQAGEDEDE EEDDEEEEDE SSSSGGGEEE SSAESLVGSS GGSSSDETRS LSPGAASSSS GDGDGKEGLE E PKGPRGSQ GGGGGGSSSS SVVSSGGDEG YGTGGGGSSA TSGGRRGSLE MSSDGEPLSR MDSEDSISST IMDVDSTISS GR STPAMMN GQGSTTSSSK NIAYNCCWDQ CQACFNSSPD LADHIRSIHV DGQRGGVFVC LWKGCKVYNT PSTSQSWLQR HML THSGDK PFKCVVGGCN ASFASQGGLA RHVPTHFSQQ NSSKVSSQPK AKEESPSKAG MNKRRKLKNK RRRSLPRPHD FFDA QTLDA IRHRAICFNL SAHIESLGKG HSVVFHSTVI AKRKEDSGKI KLLLHWMPED ILPDVWVNES ERHQLKTKVV HLSKL PKDT ALLLDPNIYR TMPQKRLKRT LIRKVFNLYL SKQ UniProtKB: Zinc finger protein AEBP2 |
-Macromolecule #3: Polycomb protein EED
| Macromolecule | Name: Polycomb protein EED / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 50.267691 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MSEREVSTAP AGTDMPAAKK QKLSSDENSN PDLSGDENDD AVSIESGTNT ERPDTPTNTP NAPGRKSWGK GKWKSKKCKY SFKCVNSLK EDHNQPLFGV QFNWHSKEGD PLVFATVGSN RVTLYECHSQ GEIRLLQSYV DADADENFYT CAWTYDSNTS H PLLAVAGS ...String: MSEREVSTAP AGTDMPAAKK QKLSSDENSN PDLSGDENDD AVSIESGTNT ERPDTPTNTP NAPGRKSWGK GKWKSKKCKY SFKCVNSLK EDHNQPLFGV QFNWHSKEGD PLVFATVGSN RVTLYECHSQ GEIRLLQSYV DADADENFYT CAWTYDSNTS H PLLAVAGS RGIIRIINPI TMQCIKHYVG HGNAINELKF HPRDPNLLLS VSKDHALRLW NIQTDTLVAI FGGVEGHRDE VL SADYDLL GEKIMSCGMD HSLKLWRINS KRMMNAIKES YDYNPNKTNR PFISQKIHFP DFSTRDIHRN YVDCVRWLGD LIL SKSCEN AIVCWKPGKM EDDIDKIKPS ESNVTILGRF DYSQCDIWYM RFSMDFWQKM LALGNQVGKL YVWDLEVEDP HKAK CTTLT HHKCGAAIRQ TSFSRDSSIL IAVCDDASIW RWDRLR UniProtKB: Polycomb protein EED |
-Macromolecule #4: Histone-lysine N-methyltransferase EZH2
| Macromolecule | Name: Histone-lysine N-methyltransferase EZH2 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO / EC number: [histone H3]-lysine27 N-trimethyltransferase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 85.492297 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MGQTGKKSEK GPVCWRKRVK SEYMRLRQLK RFRRADEVKS MFSSNRQKIL ERTEILNQEW KQRRIQPVHI LTSVSSLRGT RECSVTSDL DFPTQVIPLK TLNAVASVPI MYSWSPLQQN FMVEDETVLH NIPYMGDEVL DQDGTFIEEL IKNYDGKVHG D RECGFIND ...String: MGQTGKKSEK GPVCWRKRVK SEYMRLRQLK RFRRADEVKS MFSSNRQKIL ERTEILNQEW KQRRIQPVHI LTSVSSLRGT RECSVTSDL DFPTQVIPLK TLNAVASVPI MYSWSPLQQN FMVEDETVLH NIPYMGDEVL DQDGTFIEEL IKNYDGKVHG D RECGFIND EIFVELVNAL GQYNDDDDDD DGDDPEEREE KQKDLEDHRD DKESRPPRKF PSDKIFEAIS SMFPDKGTAE EL KEKYKEL TEQQLPGALP PECTPNIDGP NAKSVQREQS LHSFHTLFCR RCFKYDCFLH PFHATPNTYK RKNTETALDN KPC GPQCYQ HLEGAKEFAA ALTAERIKTP PKRPGGRRRG RLPNNSSRPS TPTINVLESK DTDSDREAGT ETGGENNDKE EEEK KDETS SSSEANSRCQ TPIKMKPNIE PPENVEWSGA EASMFRVLIG TYYDNFCAIA RLIGTKTCRQ VYEFRVKESS IIAPA PAED VDTPPRKKKR KHRLWAAHCR KIQLKKDGSS NHVYNYQPCD HPRQPCDSSC PCVIAQNFCE KFCQCSSECQ NRFPGC RCK AQCNTKQCPC YLAVRECDPD LCLTCGAADH WDSKNVSCKN CSIQRGSKKH LLLAPSDVAG WGIFIKDPVQ KNEFISE YC GEIISQDEAD RRGKVYDKYM CSFLFNLNND FVVDATRKGN KIRFANHSVN PNCYAKVMMV NGDHRIGIFA KRAIQTGE E LFFDYRYSQA DALKYVGIER EMEIP UniProtKB: Histone-lysine N-methyltransferase EZH2 |
-Macromolecule #5: Polycomb protein SUZ12
| Macromolecule | Name: Polycomb protein SUZ12 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 83.181922 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MAPQKHGGGG GGGSGPSAGS GGGGFGGSAA VAAATASGGK SGGGSCGGGG SYSASSSSSA AAAAGAAVLP VKKPKMEHVQ ADHELFLQA FEKPTQIYRF LRTRNLIAPI FLHRTLTYMS HRNSRTNIKR KTFKVDDMLS KVEKMKGEQE SHSLSAHLQL T FTGFFHKN ...String: MAPQKHGGGG GGGSGPSAGS GGGGFGGSAA VAAATASGGK SGGGSCGGGG SYSASSSSSA AAAAGAAVLP VKKPKMEHVQ ADHELFLQA FEKPTQIYRF LRTRNLIAPI FLHRTLTYMS HRNSRTNIKR KTFKVDDMLS KVEKMKGEQE SHSLSAHLQL T FTGFFHKN DKPSPNSENE QNSVTLEVLL VKVCHKKRKD VSCPIRQVPT GKKQVPLNPD LNQTKPGNFP SLAVSSNEFE PS NSHMVKS YSLLFRVTRP GRREFNGMIN GETNENIDVN EELPARRKRN REDGEKTFVA QMTVFDKNRR LQLLDGEYEV AMQ EMEECP ISKKRATWET ILDGKRLPPF ETFSQGPTLQ FTLRWTGETN DKSTAPIAKP LATRNSESLH QENKPGSVKP TQTI AVKES LTTDLQTRKE KDTPNENRQK LRIFYQFLYN NNTRQQTEAR DDLHCPWCTL NCRKLYSLLK HLKLCHSRFI FNYVY HPKG ARIDVSINEC YDGSYAGNPQ DIHRQPGFAF SRNGPVKRTP ITHILVCRPK RTKASMSEFL ESEDGEVEQQ RTYSSG HNR LYFHSDTCLP LRPQEMEVDS EDEKDPEWLR EKTITQIEEF SDVNEGEKEV MKLWNLHVMK HGFIADNQMN HACMLFV EN YGQKIIKKNL CRNFMLHLVS MHDFNLISIM SIDKAVTKLR EMQQKLEKGE SASPANEEIT EEQNGTANGF SEINSKEK A LETDSVSGVS KQSKKQKL UniProtKB: Polycomb protein SUZ12 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2.4 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
Details: 200 mM NaCl, 20 mM HEPES pH 7.5, 1 mM TCEP, 0.01% NP-40 | |||||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 200 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 120 sec. / Pretreatment - Atmosphere: AIR / Details: 0.24 mBar, 120 s, 10 mAmp | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV / Details: Blotting 3 seconds. | |||||||||||||||
| Details | Prior to cryo-EM sample preparation, complexes were PEGylated at 0.9 mg/mL with 5 mM MS(PEG)4 Methyl-PEG-NHS-Ester (ThermoFisher Scientific) for 2 h at 4oC. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Quantum ER / Energy filter - Slit width: 10 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 105000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller





















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)