[English] 日本語
 Yorodumi
Yorodumi- EMDB-27500: EBNA1 DNA binding domain (DBD) (458-617)+2 repeats of family repe... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | EBNA1 DNA binding domain (DBD) (458-617)+2 repeats of family repeat (FR) region | ||||||||||||||||||
 Map data Map data | |||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||
 Keywords Keywords | EBNA1 / OriP / EBV / family repeats (FR) / VIRAL PROTEIN-DNA complex | ||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationhost cell PML body / viral latency / Hydrolases; Acting on ester bonds; Endodeoxyribonucleases producing 5'-phosphomonoesters / enzyme-substrate adaptor activity / symbiont-mediated disruption of host cell PML body / regulation of DNA replication / symbiont-mediated suppression of host NF-kappaB cascade / endonuclease activity / DNA-binding transcription factor activity / positive regulation of DNA-templated transcription / DNA binding Similarity search - Function | ||||||||||||||||||
| Biological species |  Human herpesvirus 4 strain B95-8 (Epstein-Barr virus (strain B95-8)) Human herpesvirus 4 strain B95-8 (Epstein-Barr virus (strain B95-8)) | ||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.23 Å | ||||||||||||||||||
 Authors Authors | Mei Y / Lieberman PM / Murakami K | ||||||||||||||||||
| Funding support |  United States, 5 items United States, 5 items
| ||||||||||||||||||
 Citation Citation |  Journal: J Virol / Year: 2022 Journal: J Virol / Year: 2022Title: Cryo-EM Structure and Functional Studies of EBNA1 Binding to the Family of Repeats and Dyad Symmetry Elements of Epstein-Barr Virus . Authors: Yang Mei / Troy E Messick / Jayaraju Dheekollu / Hee Jong Kim / Sudheer Molugu / Leonardo Josué Castro Muñoz / Vera Moiskeenkova-Bell / Kenji Murakami / Paul M Lieberman /  Abstract: Epstein-Barr nuclear antigen 1 (EBNA1) is a multifunctional viral-encoded DNA-binding protein essential for Epstein-Barr virus (EBV) DNA replication and episome maintenance. EBNA1 binds to two ...Epstein-Barr nuclear antigen 1 (EBNA1) is a multifunctional viral-encoded DNA-binding protein essential for Epstein-Barr virus (EBV) DNA replication and episome maintenance. EBNA1 binds to two functionally distinct elements at the viral origin of plasmid replication (), termed the dyad symmetry (DS) element, required for replication initiation and the family of repeats (FR) required for episome maintenance. Here, we determined the cryo-electron microscopy (cryo-EM) structure of the EBNA1 DNA binding domain (DBD) from amino acids (aa) 459 to 614 and its interaction with two tandem sites at the DS and FR. We found that EBNA1 induces a strong DNA bending angle in the DS, while the FR is more linear. The N-terminal arm of the DBD (aa 444 to 468) makes extensive contact with DNA as it wraps around the minor groove, with some conformational variation among EBNA1 monomers. Mutation of variable-contact residues K460 and K461 had only minor effects on DNA binding but had abrogated -dependent DNA replication. We also observed that the AT-rich intervening DNA between EBNA1 binding sites in the FR can be occupied by the EBNA1 AT hook, N-terminal domain (NTD) aa 1 to 90 to form a Zn-dependent stable complex with EBNA1 DBD on a 2×FR DNA template. We propose a model showing EBNA1 DBD and NTD cobinding at the FR and suggest that this may contribute to the oligomerization of viral episomes important for maintenance during latent infection. EBV latent infection is causally linked to diverse cancers and autoimmune disorders. EBNA1 is the viral-encoded DNA binding protein required for episomal maintenance during latent infection and is consistently expressed in all EBV tumors. The interaction of EBNA1 with different genetic elements confers different viral functions, such as replication initiation at DS and chromosome tethering at FR. Here, we used cryo-EM to determine the structure of the EBNA1 DNA-binding domain (DBD) bound to two tandem sites at the DS and at the FR. We also show that the NTD of EBNA1 can interact with the AT-rich DNA sequence between tandem EBNA1 DBD binding sites in the FR. These results provide new information on the mechanism of EBNA1 DNA binding at DS and FR and suggest a higher-order oligomeric structure of EBNA1 bound to FR. Our findings have implications for targeting EBNA1 in EBV-associated disease. | ||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_27500.map.gz emd_27500.map.gz | 4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-27500-v30.xml emd-27500-v30.xml emd-27500.xml emd-27500.xml | 19 KB 19 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_27500.png emd_27500.png | 38.1 KB | ||
| Filedesc metadata |  emd-27500.cif.gz emd-27500.cif.gz | 5.9 KB | ||
| Others |  emd_27500_half_map_1.map.gz emd_27500_half_map_1.map.gz emd_27500_half_map_2.map.gz emd_27500_half_map_2.map.gz | 39.6 MB 39.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-27500 http://ftp.pdbj.org/pub/emdb/structures/EMD-27500 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27500 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27500 | HTTPS FTP |
-Validation report
| Summary document |  emd_27500_validation.pdf.gz emd_27500_validation.pdf.gz | 608.4 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_27500_full_validation.pdf.gz emd_27500_full_validation.pdf.gz | 608 KB | Display | |
| Data in XML |  emd_27500_validation.xml.gz emd_27500_validation.xml.gz | 11.4 KB | Display | |
| Data in CIF |  emd_27500_validation.cif.gz emd_27500_validation.cif.gz | 13.3 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27500 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27500 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27500 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-27500 | HTTPS FTP |
-Related structure data
| Related structure data |  8dlfMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_27500.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_27500.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.0694 Å | ||||||||||||||||||||||||||||||||||||
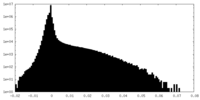
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_27500_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
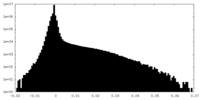
| Density Histograms |
-Half map: #2
| File | emd_27500_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
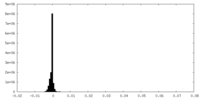
| Density Histograms |
- Sample components
Sample components
-Entire : EBNA1 DBD+2XFR complex
| Entire | Name: EBNA1 DBD+2XFR complex |
|---|---|
| Components |
|
-Supramolecule #1: EBNA1 DBD+2XFR complex
| Supramolecule | Name: EBNA1 DBD+2XFR complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Human herpesvirus 4 strain B95-8 (Epstein-Barr virus (strain B95-8)) Human herpesvirus 4 strain B95-8 (Epstein-Barr virus (strain B95-8)) |
-Macromolecule #1: Epstein-Barr nuclear antigen 1
| Macromolecule | Name: Epstein-Barr nuclear antigen 1 / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Human herpesvirus 4 strain B95-8 (Epstein-Barr virus (strain B95-8)) Human herpesvirus 4 strain B95-8 (Epstein-Barr virus (strain B95-8))Strain: B95-8 |
| Molecular weight | Theoretical: 17.564369 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: RRKKGGWFGK HRGQGGSNPK FENIAEGLRA LLARSHVERT TDEGTWVAGV FVYGGSKTSL YNLRRGTALA IPQCRLTPLS RLPFGMAPG PGPQPGPLRE SIVCYFMVFL QTHIFAEVLK DAIKDLVMTK PAPTCNIRVT VCSFDDGVDL PPWFPPMVEG A UniProtKB: Epstein-Barr nuclear antigen 1 |
-Macromolecule #2: 2XFR DNA (56-MER)
| Macromolecule | Name: 2XFR DNA (56-MER) / type: dna / ID: 2 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  Human herpesvirus 4 strain B95-8 (Epstein-Barr virus (strain B95-8)) Human herpesvirus 4 strain B95-8 (Epstein-Barr virus (strain B95-8)) |
| Molecular weight | Theoretical: 17.241064 KDa |
| Sequence | String: (DA)(DT)(DC)(DT)(DG)(DG)(DG)(DT)(DA)(DG) (DT)(DA)(DT)(DA)(DT)(DG)(DC)(DT)(DA)(DT) (DC)(DC)(DT)(DA)(DA)(DT)(DT)(DT)(DA) (DT)(DA)(DT)(DC)(DT)(DG)(DG)(DG)(DT)(DA) (DG) (DC)(DA)(DT)(DA)(DG)(DG) ...String: (DA)(DT)(DC)(DT)(DG)(DG)(DG)(DT)(DA)(DG) (DT)(DA)(DT)(DA)(DT)(DG)(DC)(DT)(DA)(DT) (DC)(DC)(DT)(DA)(DA)(DT)(DT)(DT)(DA) (DT)(DA)(DT)(DC)(DT)(DG)(DG)(DG)(DT)(DA) (DG) (DC)(DA)(DT)(DA)(DG)(DG)(DC)(DT) (DA)(DT)(DC)(DC)(DT)(DA)(DT)(DC) |
-Macromolecule #3: 2XFR DNA (56-MER)
| Macromolecule | Name: 2XFR DNA (56-MER) / type: dna / ID: 3 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  Human herpesvirus 4 strain B95-8 (Epstein-Barr virus (strain B95-8)) Human herpesvirus 4 strain B95-8 (Epstein-Barr virus (strain B95-8)) |
| Molecular weight | Theoretical: 17.264139 KDa |
| Sequence | String: (DG)(DA)(DT)(DA)(DG)(DG)(DA)(DT)(DA)(DG) (DC)(DC)(DT)(DA)(DT)(DG)(DC)(DT)(DA)(DC) (DC)(DC)(DA)(DG)(DA)(DT)(DA)(DT)(DA) (DA)(DA)(DT)(DT)(DA)(DG)(DG)(DA)(DT)(DA) (DG) (DC)(DA)(DT)(DA)(DT)(DA) ...String: (DG)(DA)(DT)(DA)(DG)(DG)(DA)(DT)(DA)(DG) (DC)(DC)(DT)(DA)(DT)(DG)(DC)(DT)(DA)(DC) (DC)(DC)(DA)(DG)(DA)(DT)(DA)(DT)(DA) (DA)(DA)(DT)(DT)(DA)(DG)(DG)(DA)(DT)(DA) (DG) (DC)(DA)(DT)(DA)(DT)(DA)(DC)(DT) (DA)(DC)(DC)(DC)(DA)(DG)(DA)(DT) |
-Macromolecule #4: water
| Macromolecule | Name: water / type: ligand / ID: 4 / Number of copies: 4 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
| ||||||||||||
| Grid | Model: C-flat-1.2/1.3 / Material: COPPER / Mesh: 300 | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 22 % / Chamber temperature: 293 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number grids imaged: 1 / Number real images: 5300 / Average exposure time: 4.0 sec. / Average electron dose: 1.25 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Calibrated defocus max: 3.0 µm / Calibrated defocus min: 10.0 µm / Calibrated magnification: 105000 / Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 105000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.23 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 1242375 |
| Initial angle assignment | Type: NOT APPLICABLE |
| Final angle assignment | Type: NOT APPLICABLE |
-Atomic model buiding 1
| Refinement | Protocol: AB INITIO MODEL |
|---|---|
| Output model |  PDB-8dlf: |
 Movie
Movie Controller
Controller



 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)