[English] 日本語
 Yorodumi
Yorodumi- EMDB-17763: Structure of the human nuclear cap-binding complex bound to ARS2[... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the human nuclear cap-binding complex bound to ARS2[147-871] and m7GTP | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Nuclear cap-binding complex / ARS2 / Pol II transcript metabolism / RNA BINDING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationmRNA cap binding complex binding / positive regulation of RNA binding / snRNA export from nucleus / nuclear cap binding complex / mRNA metabolic process / RNA cap binding complex / histone mRNA metabolic process / positive regulation of mRNA 3'-end processing / positive regulation of RNA export from nucleus / cap-dependent translational initiation ...mRNA cap binding complex binding / positive regulation of RNA binding / snRNA export from nucleus / nuclear cap binding complex / mRNA metabolic process / RNA cap binding complex / histone mRNA metabolic process / positive regulation of mRNA 3'-end processing / positive regulation of RNA export from nucleus / cap-dependent translational initiation / Processing of Intronless Pre-mRNAs / snRNA binding / RNA cap binding / alternative mRNA splicing, via spliceosome / neuronal stem cell population maintenance / miRNA-mediated post-transcriptional gene silencing / primary miRNA processing / regulation of mRNA processing / SLBP independent Processing of Histone Pre-mRNAs / SLBP Dependent Processing of Replication-Dependent Histone Pre-mRNAs / regulatory ncRNA-mediated post-transcriptional gene silencing / Transport of the SLBP independent Mature mRNA / Transport of the SLBP Dependant Mature mRNA / RNA 7-methylguanosine cap binding / response to arsenic-containing substance / Transport of Mature mRNA Derived from an Intronless Transcript / mRNA 3'-end processing / mRNA 3'-end processing / RNA catabolic process / mRNA cis splicing, via spliceosome / Transport of Mature mRNA derived from an Intron-Containing Transcript / Abortive elongation of HIV-1 transcript in the absence of Tat / positive regulation of mRNA splicing, via spliceosome / regulation of translational initiation / FGFR2 alternative splicing / RNA Polymerase II Transcription Termination / nuclear-transcribed mRNA catabolic process, nonsense-mediated decay / positive regulation of neurogenesis / Signaling by FGFR2 IIIa TM / Formation of the Early Elongation Complex / Formation of the HIV-1 Early Elongation Complex / mRNA Capping / spliceosomal complex assembly / mRNA Splicing - Minor Pathway / Processing of Capped Intron-Containing Pre-mRNA / RNA polymerase II transcribes snRNA genes / 7-methylguanosine mRNA capping / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / Formation of HIV-1 elongation complex containing HIV-1 Tat / Formation of HIV elongation complex in the absence of HIV Tat / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / mRNA export from nucleus / positive regulation of transcription elongation by RNA polymerase II / Formation of RNA Pol II elongation complex / RNA Polymerase II Pre-transcription Events / mRNA Splicing - Major Pathway / RNA splicing / mRNA transcription by RNA polymerase II / Regulation of expression of SLITs and ROBOs / mRNA splicing, via spliceosome / mRNA processing / positive regulation of cell growth / protein-macromolecule adaptor activity / snRNP Assembly / defense response to virus / molecular adaptor activity / nuclear body / ribonucleoprotein complex / mRNA binding / regulation of DNA-templated transcription / protein-containing complex / mitochondrion / DNA binding / RNA binding / nucleoplasm / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
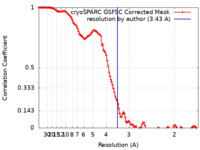
| Method | single particle reconstruction / cryo EM / Resolution: 3.43 Å | |||||||||
 Authors Authors | Dubiez E / Pellegrini E / Foucher AE / Cusack S / Kadlec J | |||||||||
| Funding support |  France, 1 items France, 1 items
| |||||||||
 Citation Citation |  Journal: Cell Rep / Year: 2024 Journal: Cell Rep / Year: 2024Title: Structural basis for competitive binding of productive and degradative co-transcriptional effectors to the nuclear cap-binding complex. Authors: Etienne Dubiez / Erika Pellegrini / Maja Finderup Brask / William Garland / Anne-Emmanuelle Foucher / Karine Huard / Torben Heick Jensen / Stephen Cusack / Jan Kadlec /   Abstract: The nuclear cap-binding complex (CBC) coordinates co-transcriptional maturation, transport, or degradation of nascent RNA polymerase II (Pol II) transcripts. CBC with its partner ARS2 forms mutually ...The nuclear cap-binding complex (CBC) coordinates co-transcriptional maturation, transport, or degradation of nascent RNA polymerase II (Pol II) transcripts. CBC with its partner ARS2 forms mutually exclusive complexes with diverse "effectors" that promote either productive or destructive outcomes. Combining AlphaFold predictions with structural and biochemical validation, we show how effectors NCBP3, NELF-E, ARS2, PHAX, and ZC3H18 form competing binary complexes with CBC and how PHAX, NCBP3, ZC3H18, and other effectors compete for binding to ARS2. In ternary CBC-ARS2 complexes with PHAX, NCBP3, or ZC3H18, ARS2 is responsible for the initial effector recruitment but inhibits their direct binding to the CBC. We show that in vivo ZC3H18 binding to both CBC and ARS2 is required for nuclear RNA degradation. We propose that recruitment of PHAX to CBC-ARS2 can lead, with appropriate cues, to competitive displacement of ARS2 and ZC3H18 from the CBC, thus promoting a productive rather than a degradative RNA fate. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17763.map.gz emd_17763.map.gz | 89.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17763-v30.xml emd-17763-v30.xml emd-17763.xml emd-17763.xml | 17.8 KB 17.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_17763_fsc.xml emd_17763_fsc.xml | 11.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_17763.png emd_17763.png | 34 KB | ||
| Masks |  emd_17763_msk_1.map emd_17763_msk_1.map | 178 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-17763.cif.gz emd-17763.cif.gz | 6.6 KB | ||
| Others |  emd_17763_half_map_1.map.gz emd_17763_half_map_1.map.gz emd_17763_half_map_2.map.gz emd_17763_half_map_2.map.gz | 165.4 MB 165.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17763 http://ftp.pdbj.org/pub/emdb/structures/EMD-17763 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17763 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17763 | HTTPS FTP |
-Validation report
| Summary document |  emd_17763_validation.pdf.gz emd_17763_validation.pdf.gz | 941 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_17763_full_validation.pdf.gz emd_17763_full_validation.pdf.gz | 940.5 KB | Display | |
| Data in XML |  emd_17763_validation.xml.gz emd_17763_validation.xml.gz | 20.6 KB | Display | |
| Data in CIF |  emd_17763_validation.cif.gz emd_17763_validation.cif.gz | 26.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17763 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17763 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17763 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17763 | HTTPS FTP |
-Related structure data
| Related structure data |  8pmpMC  8by6C  8pntC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_17763.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17763.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.839 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_17763_msk_1.map emd_17763_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_17763_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_17763_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Structure of the human nuclear cap-binding complex bound to ARS2[...
| Entire | Name: Structure of the human nuclear cap-binding complex bound to ARS2[147-871] and m7GTP |
|---|---|
| Components |
|
-Supramolecule #1: Structure of the human nuclear cap-binding complex bound to ARS2[...
| Supramolecule | Name: Structure of the human nuclear cap-binding complex bound to ARS2[147-871] and m7GTP type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Nuclear cap-binding protein subunit 1
| Macromolecule | Name: Nuclear cap-binding protein subunit 1 / type: protein_or_peptide / ID: 1 Details: CBP80 construct has the N-terminal 20 residues (containing the NLS) deleted and replaced by a methionine Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 89.781938 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MKTSDANETE DHLESLICKV GEKSACSLES NLEGLAGVLE ADLPNYKSKI LRLLCTVARL LPEKLTIYTT LVGLLNARNY NFGGEFVEA MIRQLKESLK ANNYNEAVYL VRFLSDLVNC HVIAAPSMVA MFENFVSVTQ EEDVPQVRRD WYVYAFLSSL P WVGKELYE ...String: MKTSDANETE DHLESLICKV GEKSACSLES NLEGLAGVLE ADLPNYKSKI LRLLCTVARL LPEKLTIYTT LVGLLNARNY NFGGEFVEA MIRQLKESLK ANNYNEAVYL VRFLSDLVNC HVIAAPSMVA MFENFVSVTQ EEDVPQVRRD WYVYAFLSSL P WVGKELYE KKDAEMDRIF ANTESYLKRR QKTHVPMLQV WTADKPHPQE EYLDCLWAQI QKLKKDRWQE RHILRPYLAF DS ILCEALQ HNLPPFTPPP HTEDSVYPMP RVIFRMFDYT DDPEGPVMPG SHSVERFVIE ENLHCIIKSH WKERKTCAAQ LVS YPGKNK IPLNYHIVEV IFAELFQLPA PPHIDVMYTT LLIELCKLQP GSLPQVLAQA TEMLYMRLDT MNTTCVDRFI NWFS HHLSN FQFRWSWEDW SDCLSQDPES PKPKFVREVL EKCMRLSYHQ RILDIVPPTF SALCPANPTC IYKYGDESSN SLPGH SVAL CLAVAFKSKA TNDEIFSILK DVPNPNQDDD DDEGFSFNPL KIEVFVQTLL HLAAKSFSHS FSALAKFHEV FKTLAE SDE GKLHVLRVMF EVWRNHPQMI AVLVDKMIRT QIVDCAAVAN WIFSSELSRD FTRLFVWEIL HSTIRKMNKH VLKIQKE LE EAKEKLARQH KRRSDDDDRS SDRKDGVLEE QIERLQEKVE SAQSEQKNLF LVIFQRFIMI LTEHLVRCET DGTSVLTP W YKNCIERLQQ IFLQHHQIIQ QYMVTLENLL FTAELDPHIL AVFQQFCALQ A UniProtKB: Nuclear cap-binding protein subunit 1 |
-Macromolecule #2: Nuclear cap-binding protein subunit 2
| Macromolecule | Name: Nuclear cap-binding protein subunit 2 / type: protein_or_peptide / ID: 2 / Details: Additional GA at the N-terminus / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 18.028131 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MSGGLLKALR SDSYVELSQY RDQHFRGDNE EQEKLLKKSC TLYVGNLSFY TTEEQIYELF SKSGDIKKII MGLDKMKKTA CGFCFVEYY SRADAENAMR YINGTRLDDR IIRTDWDAGF KEGRQYGRGR SGGQVRDEYR QDYDAGRGGY GKLAQNQ UniProtKB: Nuclear cap-binding protein subunit 2 |
-Macromolecule #3: Serrate RNA effector molecule homolog
| Macromolecule | Name: Serrate RNA effector molecule homolog / type: protein_or_peptide / ID: 3 Details: ARS2 has N-terminal 147 residues deleted and replaced by a methionine Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 83.061391 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: VMKTFKEFLL SLDDSVDETE AVKRYNDYKL DFRRQQMQDF FLAHKDEEWF RSKYHPDEVG KRRQEARGAL QNRLRVFLSL METGWFDNL LLDIDKADAI VKMLDAAVIK MEGGTENDLR ILEQEEEEEQ AGKPGEPSKK EEGRAGAGLG DGERKTNDKD E KKEDGKQA ...String: VMKTFKEFLL SLDDSVDETE AVKRYNDYKL DFRRQQMQDF FLAHKDEEWF RSKYHPDEVG KRRQEARGAL QNRLRVFLSL METGWFDNL LLDIDKADAI VKMLDAAVIK MEGGTENDLR ILEQEEEEEQ AGKPGEPSKK EEGRAGAGLG DGERKTNDKD E KKEDGKQA ENDSSNDDKT KKSEGDGDKE EKKEDSEKEA KKSSKKRNRK HSGDDSFDEG SVSESESESE SGQAEEEKEE AE EALKEKE KPKEEEWEKP KDAAGLECKP RPLHKTCSLF MRNIAPNISR AEIISLCKRY PGFMRVALSE PQPERRFFRR GWV TFDRSV NIKEICWNLQ NIRLRECELS PGVNRDLTRR VRNINGITQH KQIVRNDIKL AAKLIHTLDD RTQLWASEPG TPPL PTSLP SQNPILKNIT DYLIEEVSAE EEELLGSSGG APPEEPPKEG NPAEINVERD EKLIKVLDKL LLYLRIVHSL DYYNT CEYP NEDEMPNRCG IIHVRGPMPP NRISHGEVLE WQKTFEEKLT PLLSVRESLS EEEAQKMGRK DPEQEVEKFV TSNTQE LGK DKWLCPLSGK KFKGPEFVRK HIFNKHAEKI EEVKKEVAFF NNFLTDAKRP ALPEIKPAQP PGPAQILPPG LTPGLPY PH QTPQGLMPYG QPRPPILGYG AGAVRPAVPT GGPPYPHAPY GAGRGNYDAF RGQGGYPGKP RNRMVRGDPR AIVEYRDL D APDDVDFF UniProtKB: Serrate RNA effector molecule homolog |
-Macromolecule #4: 7N-METHYL-8-HYDROGUANOSINE-5'-TRIPHOSPHATE
| Macromolecule | Name: 7N-METHYL-8-HYDROGUANOSINE-5'-TRIPHOSPHATE / type: ligand / ID: 4 / Number of copies: 1 / Formula: MGT |
|---|---|
| Molecular weight | Theoretical: 539.223 Da |
| Chemical component information | 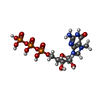 ChemComp-MGT: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 40.13 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.7000000000000001 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller
















 Z
Z Y
Y X
X