+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Rnase R bound to a 30S degradation intermediate (state II) | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | SSU / 30S / RNase R / ribosomal degradation / turnover / RIBOSOME | |||||||||
| Function / homology |  Function and homology information Function and homology informationexoribonuclease II activity / exoribonuclease II / mRNA catabolic process / ribosomal small subunit assembly / ribosomal small subunit biogenesis / small ribosomal subunit / small ribosomal subunit rRNA binding / cytosolic small ribosomal subunit / tRNA binding / rRNA binding ...exoribonuclease II activity / exoribonuclease II / mRNA catabolic process / ribosomal small subunit assembly / ribosomal small subunit biogenesis / small ribosomal subunit / small ribosomal subunit rRNA binding / cytosolic small ribosomal subunit / tRNA binding / rRNA binding / structural constituent of ribosome / ribosome / translation / ribonucleoprotein complex / response to antibiotic / mRNA binding / RNA binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.73 Å | |||||||||
 Authors Authors | Paternoga H / Dimitrova-Paternoga L / Wilson DN | |||||||||
| Funding support |  Germany, 1 items Germany, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2024 Journal: Nature / Year: 2024Title: Structural basis of ribosomal 30S subunit degradation by RNase R. Authors: Lyudmila Dimitrova-Paternoga / Sergo Kasvandik / Bertrand Beckert / Sander Granneman / Tanel Tenson / Daniel N Wilson / Helge Paternoga /     Abstract: Protein synthesis is a major energy-consuming process of the cell that requires the controlled production and turnover of ribosomes. Although the past few years have seen major advances in our ...Protein synthesis is a major energy-consuming process of the cell that requires the controlled production and turnover of ribosomes. Although the past few years have seen major advances in our understanding of ribosome biogenesis, structural insight into the degradation of ribosomes has been lacking. Here we present native structures of two distinct small ribosomal 30S subunit degradation intermediates associated with the 3' to 5' exonuclease ribonuclease R (RNase R). The structures reveal that RNase R binds at first to the 30S platform to facilitate the degradation of the functionally important anti-Shine-Dalgarno sequence and the decoding-site helix 44. RNase R then encounters a roadblock when it reaches the neck region of the 30S subunit, and this is overcome by a major structural rearrangement of the 30S head, involving the loss of ribosomal proteins. RNase R parallels this movement and relocates to the decoding site by using its N-terminal helix-turn-helix domain as an anchor. In vitro degradation assays suggest that head rearrangement poses a major kinetic barrier for RNase R, but also indicate that the enzyme alone is sufficient for complete degradation of 30S subunits. Collectively, our results provide a mechanistic basis for the degradation of 30S mediated by RNase R, and reveal that RNase R targets orphaned 30S subunits using a dynamic mechanism involving an anchored switching of binding sites. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_16596.map.gz emd_16596.map.gz | 26.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-16596-v30.xml emd-16596-v30.xml emd-16596.xml emd-16596.xml | 35.7 KB 35.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_16596.png emd_16596.png | 79.2 KB | ||
| Filedesc metadata |  emd-16596.cif.gz emd-16596.cif.gz | 8.3 KB | ||
| Others |  emd_16596_additional_1.map.gz emd_16596_additional_1.map.gz emd_16596_half_map_1.map.gz emd_16596_half_map_1.map.gz emd_16596_half_map_2.map.gz emd_16596_half_map_2.map.gz | 171.9 MB 171.8 MB 171.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-16596 http://ftp.pdbj.org/pub/emdb/structures/EMD-16596 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16596 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16596 | HTTPS FTP |
-Related structure data
| Related structure data |  8cdvMC  8cduC  8cecC  8cedC  8ceeC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_16596.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_16596.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.8 Å | ||||||||||||||||||||||||||||||||||||
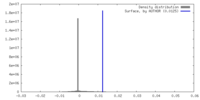
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: #1
| File | emd_16596_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
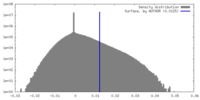
| Density Histograms |
-Half map: #1
| File | emd_16596_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
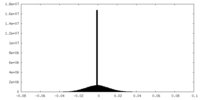
| Density Histograms |
-Half map: #2
| File | emd_16596_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
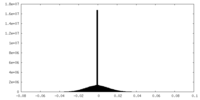
| Density Histograms |
- Sample components
Sample components
+Entire : 30S ribosomal subunit in complex with 3' exonuclease RNase R
+Supramolecule #1: 30S ribosomal subunit in complex with 3' exonuclease RNase R
+Macromolecule #1: 16S rRNA
+Macromolecule #2: RNA Substrate
+Macromolecule #3: Ribonuclease R
+Macromolecule #4: 30S ribosomal protein S4
+Macromolecule #5: 30S ribosomal protein S5
+Macromolecule #6: 30S ribosomal protein S7
+Macromolecule #7: 30S ribosomal protein S8
+Macromolecule #8: 30S ribosomal protein S9
+Macromolecule #9: 30S ribosomal protein S10
+Macromolecule #10: 30S ribosomal protein S12
+Macromolecule #11: 30S ribosomal protein S13
+Macromolecule #12: 30S ribosomal protein S15
+Macromolecule #13: 30S ribosomal protein S16
+Macromolecule #14: 30S ribosomal protein S17
+Macromolecule #15: 30S ribosomal protein S19
+Macromolecule #16: 30S ribosomal protein S20
+Macromolecule #17: 30S ribosomal protein S6
+Macromolecule #18: 30S ribosomal protein S18
+Macromolecule #19: 30S ribosomal protein S11
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE-PROPANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 0.9 µm / Nominal defocus min: 0.4 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller














 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)