+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the SEA complex (wing focused map) | |||||||||
 Map data Map data | DeepEMhancer sharpened map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | GTPase activating protein / TORC1 / Rag GTPase / SIGNALING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationurea transport / GATOR1 complex / negative regulation of small GTPase mediated signal transduction / pseudohyphal growth / Seh1-associated complex / proline transport / regulation of TORC1 signaling / regulation of autophagosome assembly / TORC1 signaling / negative regulation of TOR signaling ...urea transport / GATOR1 complex / negative regulation of small GTPase mediated signal transduction / pseudohyphal growth / Seh1-associated complex / proline transport / regulation of TORC1 signaling / regulation of autophagosome assembly / TORC1 signaling / negative regulation of TOR signaling / fungal-type vacuole membrane / vacuolar membrane / positive regulation of autophagy / signaling adaptor activity / negative regulation of TORC1 signaling / positive regulation of TORC1 signaling / cellular response to amino acid starvation / GTPase activator activity / meiotic cell cycle / protein transport / intracellular signal transduction / response to xenobiotic stimulus Similarity search - Function | |||||||||
| Biological species |  | |||||||||
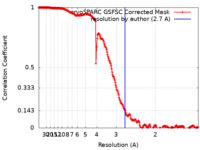
| Method | single particle reconstruction / cryo EM / Resolution: 2.7 Å | |||||||||
 Authors Authors | Tafur L / Loewith R | |||||||||
| Funding support | European Union, 2 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2022 Journal: Nature / Year: 2022Title: Cryo-EM structure of the SEA complex. Authors: Lucas Tafur / Kerstin Hinterndorfer / Caroline Gabus / Chiara Lamanna / Ariane Bergmann / Yashar Sadian / Farzad Hamdi / Fotis L Kyrilis / Panagiotis L Kastritis / Robbie Loewith /   Abstract: The SEA complex (SEAC) is a growth regulator that acts as a GTPase-activating protein (GAP) towards Gtr1, a Rag GTPase that relays nutrient status to the Target of Rapamycin Complex 1 (TORC1) in ...The SEA complex (SEAC) is a growth regulator that acts as a GTPase-activating protein (GAP) towards Gtr1, a Rag GTPase that relays nutrient status to the Target of Rapamycin Complex 1 (TORC1) in yeast. Functionally, the SEAC has been divided into two subcomplexes: SEACIT, which has GAP activity and inhibits TORC1, and SEACAT, which regulates SEACIT. This system is conserved in mammals: the GATOR complex, consisting of GATOR1 (SEACIT) and GATOR2 (SEACAT), transmits amino acid and glucose signals to mTORC1. Despite its importance, the structure of SEAC/GATOR, and thus molecular understanding of its function, is lacking. Here, we solve the cryo-EM structure of the native eight-subunit SEAC. The SEAC has a modular structure in which a COPII-like cage corresponding to SEACAT binds two flexible wings, which correspond to SEACIT. The wings are tethered to the core via Sea3, which forms part of both modules. The GAP mechanism of GATOR1 is conserved in SEACIT, and GAP activity is unaffected by SEACAT in vitro. In vivo, the wings are essential for recruitment of the SEAC to the vacuole, primarily via the EGO complex. Our results indicate that rather than being a direct inhibitor of SEACIT, SEACAT acts as a scaffold for the binding of TORC1 regulators. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_15381.map.gz emd_15381.map.gz | 883 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-15381-v30.xml emd-15381-v30.xml emd-15381.xml emd-15381.xml | 23.1 KB 23.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_15381_fsc.xml emd_15381_fsc.xml | 21.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_15381.png emd_15381.png | 64.7 KB | ||
| Masks |  emd_15381_msk_1.map emd_15381_msk_1.map | 1000 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-15381.cif.gz emd-15381.cif.gz | 8.5 KB | ||
| Others |  emd_15381_half_map_1.map.gz emd_15381_half_map_1.map.gz emd_15381_half_map_2.map.gz emd_15381_half_map_2.map.gz | 927.8 MB 927.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15381 http://ftp.pdbj.org/pub/emdb/structures/EMD-15381 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15381 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15381 | HTTPS FTP |
-Validation report
| Summary document |  emd_15381_validation.pdf.gz emd_15381_validation.pdf.gz | 1.1 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_15381_full_validation.pdf.gz emd_15381_full_validation.pdf.gz | 1.1 MB | Display | |
| Data in XML |  emd_15381_validation.xml.gz emd_15381_validation.xml.gz | 31.3 KB | Display | |
| Data in CIF |  emd_15381_validation.cif.gz emd_15381_validation.cif.gz | 41.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-15381 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-15381 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-15381 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-15381 | HTTPS FTP |
-Related structure data
| Related structure data |  8ae6MC  8adlC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_15381.map.gz / Format: CCP4 / Size: 1000 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_15381.map.gz / Format: CCP4 / Size: 1000 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | DeepEMhancer sharpened map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.726 Å | ||||||||||||||||||||||||||||||||||||
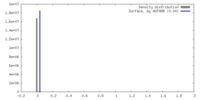
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_15381_msk_1.map emd_15381_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
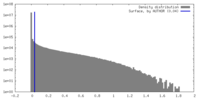
| Density Histograms |
-Half map: #2
| File | emd_15381_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
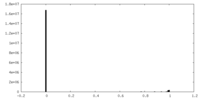
| Density Histograms |
-Half map: #1
| File | emd_15381_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
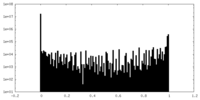
| Density Histograms |
- Sample components
Sample components
-Entire : SEAC wing module (SEACIT subcomplex)
| Entire | Name: SEAC wing module (SEACIT subcomplex) |
|---|---|
| Components |
|
-Supramolecule #1: SEAC wing module (SEACIT subcomplex)
| Supramolecule | Name: SEAC wing module (SEACIT subcomplex) / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: Reconstruction of the SEAC wing with signal subtracted, C2-symmetry expanded particles |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Nitrogen permease regulator 3
| Macromolecule | Name: Nitrogen permease regulator 3 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 130.141094 KDa |
| Sequence | String: MDECLPNSCL LGVHLVISTH SGPQIVYHYP PSNTAFLTNN PTKHQHLYGN HANLNKNTST NKEEKLFNSG STKTASQIAL NESAKSYNT AITPSMTNTN TNNVTLPPTR SHANTVGSQS SIPAATNGVG YRKTDIEDTS RTFQYQETES ETSSSGLSDS E LSTDYLDI ...String: MDECLPNSCL LGVHLVISTH SGPQIVYHYP PSNTAFLTNN PTKHQHLYGN HANLNKNTST NKEEKLFNSG STKTASQIAL NESAKSYNT AITPSMTNTN TNNVTLPPTR SHANTVGSQS SIPAATNGVG YRKTDIEDTS RTFQYQETES ETSSSGLSDS E LSTDYLDI SSDSFSISSS LSSSSLSSSP SSSSSSSPPQ DGLSRTNSSF QSTDSMSPTS PQMIMENDSI SVAESYLDSG TN NKSRAAS KRSQNFFHKL STKKSTDSKT HSPVRKLKSK PSQSTKKGNK LLKNTSNETD GNAFTGSCSI SSKKSLSSTG EHN QELRNS SLNDTPGQSP HHYHHRYHHY HKNAATSQRN SHTQYDVEEE DMEVSAMLQD GKISMNEIFF EEENFQDINK ILEF DNDFV AEFCSPEREM CNTRFEFTVD NFCFLGLPIH VDSQGRWRKS KHKNKTRSKR SSSTTTNISR KKSIASKISS LSENT LKKV NSGEADTVYD SNIGHEASTD TPNLRINTDV SGNEFEREKE DLGKNMNMFH VCFVMNPHLI EYNKRIDDMY QFVVTR LSL LLRYVQSKTS YISSECHIIL KEKERVLKHS KTYQSIRGAG NKGKYLYQRI LAKSSLARAL TECVDKIQRN EIACLEI ND DKVISLQIPI QNEFEKMPNF KLQPVLRGSY LTSILNMKFL EKSSLRIESQ NRQNDQAQFS DTNNNIYRFG NNINSTGH C GAANVDDGDD NESNYYCDDN DDLLNYALLL LDEPNNIISS LETFSYQDDI GTIILKHLVR NIQPNIPLRS YRYLITDLL DNPSSLDDLT TETNSLESSI LRSCALHLMY WRHARIVIPL SSKYTYIVSP LAPIQGYTID DYKSTSQNDG NVKKMDDREN NKSGSDRVP LIYQNSMLFR SKFPSLPSLP IFLSLLSTDK PQAYSNIIPS REHKPVYLNA LAWLIQYGYV TQLLTFINIR V DKHIKMAV DEDLEKEGFR KTNTARRPSM DYKKTDKKLD DEDGQSRDAN ASEACSGKNE GMQSNDNNKD VDEKDNENDS RV DDRDDNE IAIADEEEIL HFEYDDPEMQ HDYTIILEPE RATAIEKRWL YRCIYGQPSD IQILFNKLLK YFNGKVPMEL VII KEEISR HDLKKLLNAL DKYLIEIHHW UniProtKB: Nitrogen permease regulator 3 |
-Macromolecule #2: Maintenance of telomere capping protein 5
| Macromolecule | Name: Maintenance of telomere capping protein 5 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 131.104062 KDa |
| Sequence | String: MCSSINEGPY NSPTFGKSLS LKVDGGFNAV SINPSGRDIV LASRQGLYII DLDDPFTPPR WLHHITPWQV ADVQWSPHPA KPYWIVSTS NQKAIIWNLA KSSSNAIEFV LHGHSRAITD INFNPQHPDV LATCSVDTYV HAWDMRSPHR PFYSTSSWRS A ASQVKWNY ...String: MCSSINEGPY NSPTFGKSLS LKVDGGFNAV SINPSGRDIV LASRQGLYII DLDDPFTPPR WLHHITPWQV ADVQWSPHPA KPYWIVSTS NQKAIIWNLA KSSSNAIEFV LHGHSRAITD INFNPQHPDV LATCSVDTYV HAWDMRSPHR PFYSTSSWRS A ASQVKWNY KDPNVLASSH GNDIFVWDLR KGSTPLCSLK GHVSSVNSID FNRFKYSEIM SSSNDGTVKF WDYSKSTTES KR TVTTNFP IWRGRYLPFG EGYCIMPMVG GNNAVYLINL CDDDDSEQNK KTKLQPIYAF KGHSDRVIDF LWRSRHTCDG DYD DREFQL VTWSKDCDLK LWPISDSIYG KVNFDRGKRL EEKLPDYDYC SYNKEPENRE NVQKNEFRRL RENFVTTSGL KKNK TNHIT WLSGIRMNSA TSQEDLFNET KIQNLGEEVS AIGHKFPKVV FEKISVSTRE LCLTLNGPWS EENPDDYIFL RISIN FPLN YPNKGDPPKF TIEENSNLTM SKRQEILSNL ATIGQKYTDS NLYCLEPCIR FVLGEKVSLE DIEEGQEPLL NFDIAD HID FEELSSLDSS YSDSQNPENL SSQSDIESYK EALVFPDTSN QGLDFGRNLA LDTTPVPNGC GSCWTATGEL FCFFANE KK PEKKQNAIIK LSQKEAGVEK HPFKIEPQVL YDKEVDSSVI TAADELKARP KRYVDTLGLG GGTNGDSRTY FDDETSSD D SFDSVADDWD DILRNDIIVR TKIPILRGNF KAFSSVHSES GKTVESTKKN KNLVISKNFS SLLSDRKELA LEYLFMDAT PEGFARNNAL VAEKFDLDEI SHCWQILSDM LIDQSDYDPY TTIWNNHPMG IKWFIKEAIV YFERQQNLQM LAMLCCVILS ARRKKIPAR YYGQELENME GTIVFNDNES QNTSFWKGSD AFSTRSRSST VTPNFYGNHL RGKNIHGGDN SSIRSDDHHA R LRTHNTLN GSSKFTEPAQ KQGSRAISSS PFHSRMPDIK VELLHDDIIE AYEQEDLLHL EVSDIPKFQT YIYQYSKLLF RW GLPLERV KILKVSTDFR SSYSSQGIPP NNNKKSPYNG VLTHWIENNE FGEEKFLARN CNYCDLRVTR SSFICGNCQH VLH SSCARI WWEIGDECPS GCGCNCPEMF DA UniProtKB: Maintenance of telomere capping protein 5 |
-Macromolecule #3: Vacuolar membrane-associated protein IML1
| Macromolecule | Name: Vacuolar membrane-associated protein IML1 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 182.203359 KDa |
| Sequence | String: MFAKLHGKKQ RPISSINSQT PRTSNTTHAN SISLSSGNLI VGSNRNLRQK KEQFGSQQRA SGRKLISNKE NDDNVNNGGD NNYDNGERV HRHHIPGLKI KAYQAELGYH ESRFSENLVM LNLVEFPDIK PGDLVELKTY HKNPSASNGD KKIYFIAKDF D GETKRRAK ...String: MFAKLHGKKQ RPISSINSQT PRTSNTTHAN SISLSSGNLI VGSNRNLRQK KEQFGSQQRA SGRKLISNKE NDDNVNNGGD NNYDNGERV HRHHIPGLKI KAYQAELGYH ESRFSENLVM LNLVEFPDIK PGDLVELKTY HKNPSASNGD KKIYFIAKDF D GETKRRAK TSNVSILSGQ LQTLLDLPSR SRIWIKLKPN KFDLQADVVE FNIKDCLLNR GDMWVLSSKL VDTCVFMDQR LA FLDSIRG TIKGIYRNGK KIVSGYIGEQ TRIIFRSESA RLIFLIQITD EMWNFEETGE QLFQKMVNSF FPKIFKKWKD VDT HHTITI AFAISMDLSD TSFKDLTPGE SLKNSQDYFR IVVDQVSIIH WVDIMETLRE EFMEIRKDLL NKQTDKGYSV ANGR FSPVI KSNFLELVNF ATTILTDPFK QLDLRHTTTH VMIISPGSGL FDVDYSLLRL TGKKLLSLEM TMDLICLSKA PLHIV PLFR YRDFENKLHH CVPLWLSVFF WNDHDKKSNS EWTPRCKIYD LQMMGITENE LIREVDVEYL QLNKKVKSLS EFMNDY DKN AFEVKILCAG SNTKQSKKLN SKFDTVFEND VVVKARKIPA TATTTHGNTK FIWRGPKVAL PAIKDIQKPN VIPDLSI KT IEASFYDDCN TTNDKISTPT TSNNDNLEMN DSLVSVRSAD NQNTSLALDS LKGLSKRNSL KDFTQRVITK FISNIDTS K NKKIKSTLLR DDVDNSPLGS NTPLPSSESK ISGLKLQQKG LADENVISKR GNLIIKKNLS IFGLPSNEIM SGSPSSYLG SSHTRTSSKL SNMSDKAAFI TEGQKSKHDD SNTYSLTQQL KHRISETWVD IKSPSIPVSS EFANELLPIR WKDVWPKYVA RKYSKWRSF TTPAELPITI SDFPSKDDFD RNFIFRNHSV TLNTDQEQYN QTYKDLLRDM IYMRLLTGFQ ICVGRQVEKI E LSRESGES ETVVNKYLDF NQNDAFKLYL MIDSEIHRIT CSSSGIIDVE RYLRKDEANL FDQVPSYIPL VKTRYESSFR DA MIDPLHV KRESLNWNQI DQVLAGYGDN LIDRKWHGFR AKYVVLPTDI PPNTYSMVIN GKSETLNPEE IRVEGLRRLI GSI TRSRLR TEKEKKGRKT KREEIQPEVM FYTGPLYNFI NEQQTSLESS AINFKDSIFV NDNNLLNRNV ELSKLAYQIQ RGED RITLV NRKWHWKKHE KCFVGSEMVN WLIRNFSDID TREDAIKYGQ KVMKEGLFVH VLNKHNFLDG HYFYQFSPEY VMDTN KLEK TNSHRSTLSD PKQMLRKAST GSSNDPSAMT PFSSVVPAIS ASNASVADAK EPSRPILMLS NSLVIDVDPA GKSSKQ ESC TVHYDRVHNP DHCFHIRLEW LTTTPKLIDD LVGNWSRLCE RYGLKMIEIP WEELCTIPSV NPFHSFVEIK LAINPWE DP EFKDRELFAK SKFYYHVYLL KASGFLLDNR ASKFLQNQDI EFDIMYSWGK PQFKYVQYIH HTGAYVAELR ENGCLFLA P NNIYISRVNP GNIIGKIHSA SSSSLDAQKV ILNFKSTCLD YQKLRSIFLD AKEMWITGKI VED UniProtKB: Vacuolar membrane-associated protein IML1 |
-Macromolecule #4: Nitrogen permease regulator 2
| Macromolecule | Name: Nitrogen permease regulator 2 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 69.937547 KDa |
| Sequence | String: MLSYFQGFVP IHTIFYSVFH PTEGSKIKYE FPPNNLKNHG INFNTFKNYI IPKPILCHKL ITFKYGTYRI VCYPVTINSP IYARNFFSF NFVFVFPYDC ETSPYEPAIT RLGKMFKVLE EQNQLLSKSE RDPVFFDLKV LENSTTTPST AGPSSTPNPS S NTTPTHPT ...String: MLSYFQGFVP IHTIFYSVFH PTEGSKIKYE FPPNNLKNHG INFNTFKNYI IPKPILCHKL ITFKYGTYRI VCYPVTINSP IYARNFFSF NFVFVFPYDC ETSPYEPAIT RLGKMFKVLE EQNQLLSKSE RDPVFFDLKV LENSTTTPST AGPSSTPNPS S NTTPTHPT SEKDTKDMRS SRYSDLIKDL GLPQSAFSIQ DLLMRIFQDL NNYSECLIPI DEGNAVDIKI FPLLRPPTTC VS LEDVPLS SVNLKKIIDV NWDPTMMSIV PYIDGLNSIA KISKLSNSDP GLVIECIRHL IYYKCVTLSD IFQFSNIYAP SSL IRNFLT DPLMASDCQS YVTFPEVSKI SNLPLNKSLG SGDQDSPSFS VRRKSKSSSI PSNPDSRTTS FSSTSRVSQN SSLN SSFSS IYKDWRQSQT SCSSSNIHVI NNRNRFLPTR SCLFDLYRSL SQGQTLKTWY ESKYMILKEN NIDIRRFITF GLEKR IIYR CYSFPVMINA GSREPKEMTP IITKDLVNND KLLEKRNHNH LLSATGSRNT AQSGNLKPER PSKVSFEMQR VSSLAT GKS TMPKLSDEEE GILEESIRNA ETFDKICVLL SKPKLEVESY LNELGEFKVI NS UniProtKB: Nitrogen permease regulator 2 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: TFS Selectris X / Energy filter - Slit width: 10 eV |
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.6 µm / Nominal defocus min: 0.6 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL |
|---|---|
| Output model |  PDB-8ae6: |
 Movie
Movie Controller
Controller








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)