+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Structure of the autoinducer-2 exporter TqsA from E. coli | |||||||||
 マップデータ マップデータ | C5 symmetry imposed final map for TqsA | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | cryo-EM / membrane protein / autoinducer-2 exporter / quorum sensing / pentamer / protein oligomerization / STRUCTURAL PROTEIN | |||||||||
| 機能・相同性 | autoinducer AI-2 transmembrane transport / Transmembrane protein TqsA-like / AI-2E family transporter / quorum sensing / plasma membrane => GO:0005886 / efflux transmembrane transporter activity / transmembrane transport / AI-2 transport protein TqsA 機能・相同性情報 機能・相同性情報 | |||||||||
| 生物種 |   | |||||||||
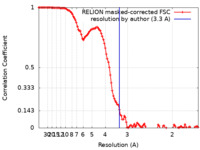
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.3 Å | |||||||||
 データ登録者 データ登録者 | Khera R / Xie H | |||||||||
| 資金援助 |  ドイツ, 1件 ドイツ, 1件
| |||||||||
 引用 引用 |  ジャーナル: EMBO J / 年: 2022 ジャーナル: EMBO J / 年: 2022タイトル: Cryo-EM structures of pentameric autoinducer-2 exporter from Escherichia coli reveal its transport mechanism. 著者: Radhika Khera / Ahmad R Mehdipour / Jani R Bolla / Joerg Kahnt / Sonja Welsch / Ulrich Ermler / Cornelia Muenke / Carol V Robinson / Gerhard Hummer / Hao Xie / Hartmut Michel /    要旨: Bacteria utilize small extracellular molecules to communicate in order to collectively coordinate their behaviors in response to the population density. Autoinducer-2 (AI-2), a universal molecule for ...Bacteria utilize small extracellular molecules to communicate in order to collectively coordinate their behaviors in response to the population density. Autoinducer-2 (AI-2), a universal molecule for both intra- and inter-species communication, is involved in the regulation of biofilm formation, virulence, motility, chemotaxis, and antibiotic resistance. While many studies have been devoted to understanding the biosynthesis and sensing of AI-2, very little information is available on its export. The protein TqsA from Escherichia coli, which belongs to the AI-2 exporter superfamily, has been shown to export AI-2. Here, we report the cryogenic electron microscopic structures of two AI-2 exporters (TqsA and YdiK) from E. coli at 3.35 Å and 2.80 Å resolutions, respectively. Our structures suggest that the AI-2 exporter exists as a homo-pentameric complex. In silico molecular docking and native mass spectrometry experiments were employed to demonstrate the interaction between AI-2 and TqsA, and the results highlight the functional importance of two helical hairpins in substrate binding. We propose that each monomer works as an independent functional unit utilizing an elevator-type transport mechanism. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_12256.map.gz emd_12256.map.gz | 11.7 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-12256-v30.xml emd-12256-v30.xml emd-12256.xml emd-12256.xml | 19 KB 19 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_12256_fsc.xml emd_12256_fsc.xml | 11.4 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_12256.png emd_12256.png | 107 KB | ||
| マスクデータ |  emd_12256_msk_1.map emd_12256_msk_1.map | 125 MB |  マスクマップ マスクマップ | |
| Filedesc metadata |  emd-12256.cif.gz emd-12256.cif.gz | 5.9 KB | ||
| その他 |  emd_12256_additional_1.map.gz emd_12256_additional_1.map.gz emd_12256_half_map_1.map.gz emd_12256_half_map_1.map.gz emd_12256_half_map_2.map.gz emd_12256_half_map_2.map.gz | 13.5 MB 98.3 MB 98.3 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12256 http://ftp.pdbj.org/pub/emdb/structures/EMD-12256 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12256 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12256 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_12256_validation.pdf.gz emd_12256_validation.pdf.gz | 765 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_12256_full_validation.pdf.gz emd_12256_full_validation.pdf.gz | 764.6 KB | 表示 | |
| XML形式データ |  emd_12256_validation.xml.gz emd_12256_validation.xml.gz | 18.7 KB | 表示 | |
| CIF形式データ |  emd_12256_validation.cif.gz emd_12256_validation.cif.gz | 24.6 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12256 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12256 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12256 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12256 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  7nb6MC  7ot9C M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_12256.map.gz / 形式: CCP4 / 大きさ: 125 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_12256.map.gz / 形式: CCP4 / 大きさ: 125 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | C5 symmetry imposed final map for TqsA | ||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 0.837 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-マスク #1
| ファイル |  emd_12256_msk_1.map emd_12256_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
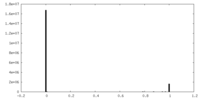
| 密度ヒストグラム |
-追加マップ: C1 symmetry map
| ファイル | emd_12256_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | C1 symmetry map | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
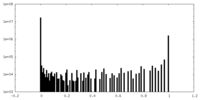
| 密度ヒストグラム |
-ハーフマップ: Half map2
| ファイル | emd_12256_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half map2 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
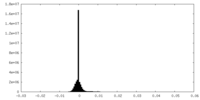
| 密度ヒストグラム |
-ハーフマップ: Half map1
| ファイル | emd_12256_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half map1 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
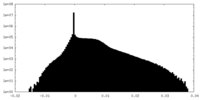
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Pentameric TqsA
| 全体 | 名称: Pentameric TqsA |
|---|---|
| 要素 |
|
-超分子 #1: Pentameric TqsA
| 超分子 | 名称: Pentameric TqsA / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 187 KDa |
-分子 #1: AI-2 transport protein TqsA
| 分子 | 名称: AI-2 transport protein TqsA / タイプ: protein_or_peptide / ID: 1 / コピー数: 5 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 37.563543 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MAKPIITLNG LKIVIMLGML VIILCGIRFA AEIIVPFILA LFIAVILNPL VQHMVRWRVP RVLAVSILMT IIVMAMVLLL AYLGSALNE LTRTLPQYRN SIMTPLQALE PLLQRVGIDV SVDQLAHYID PNAAMTLLTN LLTQLSNAMS SIFLLLLTVL F MLLEVPQL ...文字列: MAKPIITLNG LKIVIMLGML VIILCGIRFA AEIIVPFILA LFIAVILNPL VQHMVRWRVP RVLAVSILMT IIVMAMVLLL AYLGSALNE LTRTLPQYRN SIMTPLQALE PLLQRVGIDV SVDQLAHYID PNAAMTLLTN LLTQLSNAMS SIFLLLLTVL F MLLEVPQL PGKFQQMMAR PVEGMAAIQR AIDSVSHYLV LKTAISIITG LVAWAMLAAL DVRFAFVWGL LAFALNYIPN IG SVLAAIP PIAQVLVFNG FYEALLVLAG YLLINLVFGN ILEPRIMGRG LGLSTLVVFL SLIFWGWLLG PVGMLLSVPL TII VKIALE QTAGGQSIAV LLSDLNKE UniProtKB: AI-2 transport protein TqsA |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 3.4 mg/mL |
|---|---|
| 緩衝液 | pH: 7.5 / 詳細: 50 mM Tris (pH 7.5), 150 mM NaCl and 0.006% GDN |
| グリッド | モデル: C-flat-1.2/1.3 / 材質: COPPER / メッシュ: 300 / 前処理 - タイプ: GLOW DISCHARGE |
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 277.15 K / 装置: FEI VITROBOT MARK III / 詳細: blot time 4 s, blot force +20. |
| 詳細 | Its a membrane protein and for purification, glyco-diosgenin was used. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K3 BIOQUANTUM (6k x 4k) デジタル化 - サイズ - 横: 5760 pixel / デジタル化 - サイズ - 縦: 4092 pixel / 撮影したグリッド数: 1 / 実像数: 5452 / 平均電子線量: 80.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: OTHER / 撮影モード: OTHER / Cs: 2.7 mm |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER ホルダー冷却材: NITROGEN |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー





 Z
Z Y
Y X
X