+Search query
-Structure paper
| Title | Structural Elucidation of the Mechanism for Inhibitor Resistance in the Na-Translocating NADH-Ubiquinone Oxidoreductase from . |
|---|---|
| Journal, issue, pages | Biochemistry, Vol. 64, Issue 9, Page 1963-1972, Year 2025 |
| Publish date | May 6, 2025 |
 Authors Authors | Moe Ishikawa-Fukuda / Jun-Ichi Kishikawa / Takahiro Masuya / Takeshi Ito / Nicole L Butler / Danielle McFee / Takayuki Kato / Blanca Barquera / Hideto Miyoshi / Masatoshi Murai /   |
| PubMed Abstract | Na-translocating NADH-ubiquinone oxidoreductase (Na-NQR) is a unique redox-driven Na-pump. Since this enzyme is exclusively found in prokaryotes, including the human pathogens and , it is a ...Na-translocating NADH-ubiquinone oxidoreductase (Na-NQR) is a unique redox-driven Na-pump. Since this enzyme is exclusively found in prokaryotes, including the human pathogens and , it is a promising target for highly selective antibiotics. Korormicin A, a natural product, and a specific and potent inhibitor of Na-NQR, may become a lead compound for the relevant drug design. We previously showed that the G141A mutation in the NqrB subunit (NqrB-G141A) confers moderate resistance to korormicin A (about 100-fold). However, the efficiency of photoaffinity labeling of the mutant enzyme by a photoreactive korormicin derivative was the same as in the wild-type enzyme. Because of these apparently conflicting results, the molecular mechanism underlying the korormicin A-resistance remains elusive. In the present study, we determined the cryo-EM structure of the NqrB-G141A mutant in the presence of bound korormicin A, and compared it to the corresponding structure from the wild-type enzyme. The toxophoric moiety of korormicin A binds to the mutant enzyme similarly to how it binds to the wild type. However, the added bulk of the alanine-141 excludes the alkyl side chain from the binding cavity, resulting in a decrease in the binding affinity. In fact, isothermal titration calorimetry revealed that the binding affinity of korormicin to the NqrB-G141A mutant is significantly weaker compared to the wild-type. Altogether, we conclude that the inhibitory potency of korormicin A is weaker in the NqrB-G141A mutant due to the decrease in its binding affinity to the altered binding cavity. |
 External links External links |  Biochemistry / Biochemistry /  PubMed:40263754 / PubMed:40263754 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 2.68 Å |
| Structure data | EMDB-63340, PDB-9lrr: |
| Chemicals |  ChemComp-FMN: 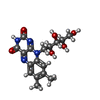 ChemComp-RBF:  ChemComp-LMT:  ChemComp-PEE: 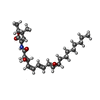 ChemComp-IQT:  ChemComp-CA:  ChemComp-FES:  ChemComp-FAD:  ChemComp-HOH: |
| Source |
|
 Keywords Keywords | MEMBRANE PROTEIN / Na+-NQR / Na+ transporter / inhibitor / oxidoreductase / drug resistant |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers





 vibrio cholerae o395 (bacteria)
vibrio cholerae o395 (bacteria)