+Search query
-Structure paper
| Title | Chaperone-assisted cryo-EM structure of P. aeruginosa PhuR reveals molecular basis for heme binding. |
|---|---|
| Journal, issue, pages | Structure, Vol. 32, Issue 4, Page 411-423.e6, Year 2024 |
| Publish date | Apr 4, 2024 |
 Authors Authors | Paweł P Knejski / Satchal K Erramilli / Anthony A Kossiakoff /   |
| PubMed Abstract | Pathogenic bacteria, such as Pseudomonas aeruginosa, depend on scavenging heme for the acquisition of iron, an essential nutrient. The TonB-dependent transporter (TBDT) PhuR is the major heme uptake ...Pathogenic bacteria, such as Pseudomonas aeruginosa, depend on scavenging heme for the acquisition of iron, an essential nutrient. The TonB-dependent transporter (TBDT) PhuR is the major heme uptake protein in P. aeruginosa clinical isolates. However, a comprehensive understanding of heme recognition and TBDT transport mechanisms, especially PhuR, remains limited. In this study, we employed single-particle cryogenic electron microscopy (cryo-EM) and a phage display-generated synthetic antibody (sAB) as a fiducial marker to enable the determination of a high-resolution (2.5 Å) structure of PhuR with a bound heme. Notably, the structure reveals iron coordination by Y529 on a conserved extracellular loop, shedding light on the role of tyrosine in heme binding. Biochemical assays and negative-stain EM demonstrated that the sAB specifically targets the heme-bound state of PhuR. These findings provide insights into PhuR's heme binding and offer a template for developing conformation-specific sABs against outer membrane proteins (OMPs) for structure-function investigations. |
 External links External links |  Structure / Structure /  PubMed:38325368 / PubMed:38325368 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 2.5 Å |
| Structure data | EMDB-41255, PDB-8the: |
| Chemicals |  ChemComp-HEM: 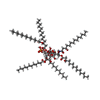 ChemComp-CTQ:  ChemComp-HOH: |
| Source |
|
 Keywords Keywords | MEMBRANE PROTEIN / beta barrel / outer membrane protein / heme binding protein / heme transport |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers






 homo sapiens (human)
homo sapiens (human)