+Search query
-Structure paper
| Title | Cryo-EM structure of cell-free synthesized human histamine 2 receptor/G complex in nanodisc environment. |
|---|---|
| Journal, issue, pages | Nat Commun, Vol. 15, Issue 1, Page 1831, Year 2024 |
| Publish date | Feb 28, 2024 |
 Authors Authors | Zoe Köck / Kilian Schnelle / Margherita Persechino / Simon Umbach / Hannes Schihada / Dovile Januliene / Kristian Parey / Steffen Pockes / Peter Kolb / Volker Dötsch / Arne Möller / Daniel Hilger / Frank Bernhard /  |
| PubMed Abstract | Here we describe the cryo-electron microscopy structure of the human histamine 2 receptor (HR) in an active conformation with bound histamine and in complex with G heterotrimeric protein at an ...Here we describe the cryo-electron microscopy structure of the human histamine 2 receptor (HR) in an active conformation with bound histamine and in complex with G heterotrimeric protein at an overall resolution of 3.4 Å. The complex was generated by cotranslational insertion of the receptor into preformed nanodisc membranes using cell-free synthesis in E. coli lysates. Structural comparison with the inactive conformation of HR and the inactive and G-coupled active state of HR together with structure-guided functional experiments reveal molecular insights into the specificity of ligand binding and G protein coupling for this receptor family. We demonstrate lipid-modulated folding of cell-free synthesized HR, its agonist-dependent internalization and its interaction with endogenously synthesized HR and HR in HEK293 cells by applying a recently developed nanotransfer technique. |
 External links External links |  Nat Commun / Nat Commun /  PubMed:38418462 / PubMed:38418462 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 3.4 Å |
| Structure data | EMDB-17793, PDB-8pok: |
| Chemicals | 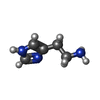 ChemComp-HSM: |
| Source |
|
 Keywords Keywords | MEMBRANE PROTEIN / GPCR / Cell-free / H2R / human histamine 2 receptor / active conformation / complex / detergent-free / lipid environment |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers





 homo sapiens (human)
homo sapiens (human)
