+Search query
-Structure paper
| Title | Natural variants modify Klebsiella pneumoniae carbapenemase (KPC) acyl-enzyme conformational dynamics to extend antibiotic resistance. |
|---|---|
| Journal, issue, pages | J. Biol. Chem., Vol. 296, Page 100126-100126, Year 2020 |
| Publish date | May 14, 2020 (structure data deposition date) |
 Authors Authors | Tooke, C.L. / Hinchliffe, P. / Bonomo, R.A. / Schofield, C.J. / Mulholland, A.J. / Spencer, J. |
 External links External links |  J. Biol. Chem. / J. Biol. Chem. /  PubMed:33257320 PubMed:33257320 |
| Methods | X-ray diffraction |
| Resolution | 1.24 - 1.4 Å |
| Structure data |  PDB-6z21:  PDB-6z22:  PDB-6z23:  PDB-6z24:  PDB-6z25: |
| Chemicals |  ChemComp-GOL:  ChemComp-SO4:  ChemComp-CL:  ChemComp-HOH: 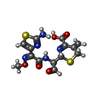 ChemComp-CEF: 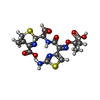 ChemComp-CAZ: |
| Source |
|
 Keywords Keywords | ANTIMICROBIAL PROTEIN / beta-lactamase / antibiotic resistance / mutant / carbapenemase. / unliganded beta-lactamase / 3-layer alpha-beta-alpha sandwich / part of the DD-peptidase and beta-lactamase superfamily. |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers



 klebsiella pneumoniae (bacteria)
klebsiella pneumoniae (bacteria)