+Search query
-Structure paper
| Title | Binding of ISRIB reveals a regulatory site in the nucleotide exchange factor eIF2B. |
|---|---|
| Journal, issue, pages | Science, Vol. 359, Issue 6383, Page 1533-1536, Year 2018 |
| Publish date | Mar 30, 2018 |
 Authors Authors | Alisa F Zyryanova / Félix Weis / Alexandre Faille / Akeel Abo Alard / Ana Crespillo-Casado / Yusuke Sekine / Heather P Harding / Felicity Allen / Leopold Parts / Christophe Fromont / Peter M Fischer / Alan J Warren / David Ron /  |
| PubMed Abstract | The integrated stress response (ISR) is a conserved translational and transcriptional program affecting metabolism, memory, and immunity. The ISR is mediated by stress-induced phosphorylation of ...The integrated stress response (ISR) is a conserved translational and transcriptional program affecting metabolism, memory, and immunity. The ISR is mediated by stress-induced phosphorylation of eukaryotic translation initiation factor 2α (eIF2α) that attenuates the guanine nucleotide exchange factor eIF2B. A chemical inhibitor of the ISR, ISRIB, reverses the attenuation of eIF2B by phosphorylated eIF2α, protecting mice from neurodegeneration and traumatic brain injury. We describe a 4.1-angstrom-resolution cryo-electron microscopy structure of human eIF2B with an ISRIB molecule bound at the interface between the β and δ regulatory subunits. Mutagenesis of residues lining this pocket altered the hierarchical cellular response to ISRIB analogs in vivo and ISRIB binding in vitro. Our findings point to a site in eIF2B that can be exploited by ISRIB to regulate translation. |
 External links External links |  Science / Science /  PubMed:29599245 / PubMed:29599245 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 4.1 Å |
| Structure data | |
| Chemicals | 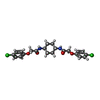 ChemComp-C7B: |
| Source |
|
 Keywords Keywords | MEMBRANE PROTEIN / GEF / Complex / ISRIB |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers





 homo sapiens (human)
homo sapiens (human)