+Search query
-Structure paper
| Title | Unprecedented access of phenolic substrates to the heme active site of a catalase: Substrate binding and peroxidase-like reactivity of Bacillus pumilus catalase monitored by X-ray crystallography and EPR spectroscopy. |
|---|---|
| Journal, issue, pages | Proteins, Vol. 83, Page 853-866, Year 2015 |
| Publish date | Jun 20, 2014 (structure data deposition date) |
 Authors Authors | Loewen, P.C. / Villanueva, J. / Switala, J. / Donald, L.J. / Ivancich, A. |
 External links External links |  Proteins / Proteins /  PubMed:25663126 PubMed:25663126 |
| Methods | X-ray diffraction |
| Resolution | 1.65 - 1.95 Å |
| Structure data |  PDB-4qol:  PDB-4qom:  PDB-4qon:  PDB-4qoo:  PDB-4qop:  PDB-4qoq:  PDB-4qor: |
| Chemicals |  ChemComp-HEM:  ChemComp-CL:  ChemComp-NA:  ChemComp-ACT:  ChemComp-HOH: 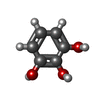 ChemComp-PYG: 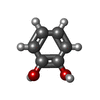 ChemComp-CAQ: 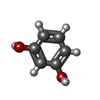 ChemComp-RCO: 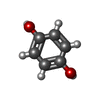 ChemComp-HQE:  ChemComp-JZ3:  ChemComp-2CH: |
| Source |
|
 Keywords Keywords | OXIDOREDUCTASE / catalase fold / catalase / peroxidase / catalase and peroxidase |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers




