+Search query
-Structure paper
| Title | Increased structural flexibility at the active site of a fluorophore-conjugated beta-lactamase distinctively impacts its binding toward diverse cephalosporin antibiotics |
|---|---|
| Journal, issue, pages | J. Biol. Chem., Vol. 286, Page 31771-31780, Year 2011 |
| Publish date | Jun 16, 2011 (structure data deposition date) |
 Authors Authors | Wong, W.-T. / Chan, K.-C. / So, P.-K. / Yap, H.-K. / Chung, W.-H. / Leung, Y.-C. / Wong, K.-Y. / Zhao, Y.-X. |
 External links External links |  J. Biol. Chem. / J. Biol. Chem. /  PubMed:21705325 PubMed:21705325 |
| Methods | X-ray diffraction |
| Resolution | 1.9 - 2.5 Å |
| Structure data |  PDB-3sh7:  PDB-3sh8:  PDB-3sh9: |
| Chemicals | 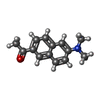 ChemComp-BB0:  ChemComp-HOH: 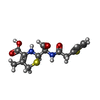 ChemComp-CED: 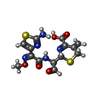 ChemComp-CEF: |
| Source |
|
 Keywords Keywords | HYDROLASE / beta-lactamase / fluorophore / beta-lactam / HYDROLASE/ANTIBIOTIC / cephaloridine / HYDROLASE-ANTIBIOTIC complex / PenP / cefotaxime |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers




