+Search query
-Structure paper
| Title | Defining AV2-1 as a novel pharmacological probe to target human and rodent TRPV2. |
|---|---|
| Journal, issue, pages | Br J Pharmacol, Year 2026 |
| Publish date | Mar 30, 2026 |
 Authors Authors | Andrea Leipe / Julia A Rocereta / Ruth A Pumroy / Andreas Leffler / Michael Schaefer / Vera Moiseenkova-Bell / Kerstin Hill /   |
| PubMed Abstract | BACKGROUND AND PURPOSE: Transient receptor potential vanilloid 2 (TRPV2) is a non-selective cation channel implicated in immune cell functions. However, progress in understanding TRPV2 has been ...BACKGROUND AND PURPOSE: Transient receptor potential vanilloid 2 (TRPV2) is a non-selective cation channel implicated in immune cell functions. However, progress in understanding TRPV2 has been limited by a lack of potent and selective pharmacological tools, particularly those targeting the human variant. We aimed to identify and characterise a novel small-molecule activator of TRPV2. EXPERIMENTAL APPROACH: We screened a compound library using Ca imaging in HEK293 cells stably expressing mouse TRPV2. The lead compound AV2-1 was validated by concentration-response analyses, microfluorometric Ca assays, and electrophysiological recordings. Structural insights were obtained from cryoEM of TRPV2 in complex with AV2-1, and mutagenesis was performed to confirm binding site residues. The efficacy of AV2-1 was assessed in human peripheral blood-derived macrophages by Ca imaging, whole-cell electrophysiology and TIRF microscopy to detect Ca microdomains. KEY RESULTS: AV2-1 is a novel TRPV2 activator showing robust efficacy across mouse, rat and human orthologues. Structural analysis reveals that AV2-1 stabilises the channel in its active conformation by binding to an established intracellular pocket via TRPV2-specific residues His165 and Cys157, as confirmed by mutagenesis experiments. AV2-1 induces TRPV2-dependent global [Ca] signals, ionic currents and localised subplasmalemmal Ca microdomains in human peripheral blood-derived macrophages. CONCLUSION AND IMPLICATIONS: AV2-1 represents a novel pharmacological tool for probing TRPV2 function in immune cells, combining improved selectivity and potency with low toxicity. Its ability to activate human TRPV2 and elicit physiologically relevant Ca signals highlights its potential for advancing TRPV2 research and for therapeutic exploration in immune modulation and disease contexts. |
 External links External links |  Br J Pharmacol / Br J Pharmacol /  PubMed:41913383 PubMed:41913383 |
| Methods | EM (single particle) |
| Resolution | 2.83 Å |
| Structure data | EMDB-48325, PDB-9mkf: |
| Chemicals |  ChemComp-PEX: 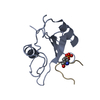 PDB-1bmb: |
| Source |
|
 Keywords Keywords | MEMBRANE PROTEIN / TRP Channel / TRPV2 |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers






