+Search query
-Structure paper
| Title | Balanced plant helper NLR activation by a modified host protein complex. |
|---|---|
| Journal, issue, pages | Nature, Vol. 639, Issue 8054, Page 447-455, Year 2025 |
| Publish date | Feb 12, 2025 |
 Authors Authors | Shijia Huang / Junli Wang / Ridan Song / Aolin Jia / Yu Xiao / Yue Sun / Lin Wang / Dennis Mahr / Zhongshou Wu / Zhifu Han / Xin Li / Jane E Parker / Jijie Chai /    |
| PubMed Abstract | Nucleotide-binding leucine-rich repeat (NLR) receptors play crucial roles in plant immunity by sensing pathogen effectors. In Arabidopsis, certain sensor NLRs function as NADases to catalyse the ...Nucleotide-binding leucine-rich repeat (NLR) receptors play crucial roles in plant immunity by sensing pathogen effectors. In Arabidopsis, certain sensor NLRs function as NADases to catalyse the production of second messengers, which can be recognized by enhanced disease susceptibility 1 (EDS1) with its partner senescence-associated gene 101 (SAG101), to activate helper NLR N requirement gene 1 (NRG1). A cryoelectron microscopy structure shows that second-messenger-activated EDS1-SAG101 mainly contacts the leucine-rich repeat domain of NRG1A to mediate the formation of an induced EDS1-SAG101-NRG1A complex. Structural comparisons show that binding of a second messenger induces conformational changes in EDS1-SAG101, which are recognized by NRG1A, leading to its allosteric activation. We further show that an inhibitory NRG1 family member, NRG1C, efficiently outcompetes NRG1A for binding to second-messenger-activated EDS1-SAG101. These findings uncover mechanisms for NRG1A activation through its recognition of a modified host EDS1-SAG101 complex, and NRG1A inhibition by NRG1C through sequestration of the activated EDS1-SAG101, thus shedding light on the activation and constraint of a central plant immune response system. |
 External links External links |  Nature / Nature /  PubMed:39939760 PubMed:39939760 |
| Methods | EM (single particle) / X-ray diffraction |
| Resolution | 2.49 - 3.09 Å |
| Structure data | EMDB-39411, PDB-8yn1:  PDB-8yn0: |
| Chemicals | 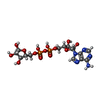 ChemComp-APR:  ChemComp-ATP:  ChemComp-HOH: |
| Source |
|
 Keywords Keywords | PLANT PROTEIN/HYDROLASE / EDS1 / SAG101 / ATP-ADPR / NRG1C / PLANT PROTEIN / PLANT PROTEIN-HYDROLASE complex / Plant immunity / ETI / NRG1A / ADPr-ATP |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers






