+Search query
-Structure paper
| Title | Structural and functional investigation of ABC transporter STE6-2p from reveals unexpected interaction with sterol molecules. |
|---|---|
| Journal, issue, pages | Proc Natl Acad Sci U S A, Vol. 119, Issue 43, Page e2202822119, Year 2022 |
| Publish date | Oct 25, 2022 |
 Authors Authors | E Sabine M Schleker / Sabine Buschmann / Hao Xie / Sonja Welsch / Hartmut Michel / Christoph Reinhart /  |
| PubMed Abstract | Adenosine triphosphate (ATP)-binding cassette (ABC) transporters are multidomain transmembrane proteins, which facilitate the transport of various substances across cell membranes using energy ...Adenosine triphosphate (ATP)-binding cassette (ABC) transporters are multidomain transmembrane proteins, which facilitate the transport of various substances across cell membranes using energy derived from ATP hydrolysis. They are important drug targets since they mediate decreased drug susceptibility during pharmacological treatments. For the methylotrophic yeast , a model organism that is a widely used host for protein expression, the role and function of its ABC transporters is unexplored. In this work, we investigated the ABC-B transporter STE6-2p. Functional investigations revealed that STE6-2p is capable of transporting rhodamines in vivo and is active in the presence of verapamil and triazoles in vitro. A phylogenetic analysis displays homology among multidrug resistance (MDR) transporters from pathogenic fungi to human ABC-B transporters. Further, we present high-resolution single-particle electron cryomicroscopy structures of an ABC transporter from in the apo conformation (3.1 Å) and in complex with verapamil and adenylyl imidodiphosphate (AMP-PNP) (3.2 Å). An unknown density between transmembrane helices 4, 5, and 6 in both structures suggests the presence of a sterol-binding site of unknown function. |
 External links External links |  Proc Natl Acad Sci U S A / Proc Natl Acad Sci U S A /  PubMed:36256814 / PubMed:36256814 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 3.1 - 3.2 Å |
| Structure data | EMDB-14049: Cryo-EM structure of ABC transporter STE6-2p from Pichia pastoris in apo conformation at 3.1 A resolution EMDB-14050, PDB-7qks: |
| Chemicals | 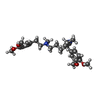 ChemComp-I6H:  ChemComp-ANP:  ChemComp-MG: 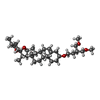 ChemComp-9Z9: |
| Source |
|
 Keywords Keywords | TRANSPORT PROTEIN / ABC transporter / ABCB / MDR / membrane protein / Pichia pastoris |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers







 komagataella phaffii cbs 7435 (fungus)
komagataella phaffii cbs 7435 (fungus)