+Search query
-Structure paper
| Title | Structure of native glycolipoprotein filaments in honeybee royal jelly. |
|---|---|
| Journal, issue, pages | Nat Commun, Vol. 11, Issue 1, Page 6267, Year 2020 |
| Publish date | Dec 8, 2020 |
 Authors Authors | Simone Mattei / Arvid Ban / Armin Picenoni / Marc Leibundgut / Rudi Glockshuber / Daniel Boehringer /   |
| PubMed Abstract | Royal jelly (RJ) is produced by honeybees (Apis mellifera) as nutrition during larval development. The high viscosity of RJ originates from high concentrations of long lipoprotein filaments that ...Royal jelly (RJ) is produced by honeybees (Apis mellifera) as nutrition during larval development. The high viscosity of RJ originates from high concentrations of long lipoprotein filaments that include the glycosylated major royal jelly protein 1 (MRJP1), the small protein apisimin and insect lipids. Using cryo-electron microscopy we reveal the architecture and the composition of RJ filaments, in which the MRJP1 forms the outer shell of the assembly, surrounding stacked apisimin tetramers harbouring tightly packed lipids in the centre. The structural data rationalize the pH-dependent disassembly of RJ filaments in the gut of the larvae. |
 External links External links |  Nat Commun / Nat Commun /  PubMed:33293513 / PubMed:33293513 /  PubMed Central PubMed Central |
| Methods | EM (helical sym.) / EM (tomography) |
| Resolution | 3.5 Å |
| Structure data | EMDB-11892, PDB-7asd:  EMDB-11898: |
| Chemicals | 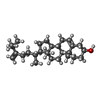 ChemComp-94R:  ChemComp-NAG:  ChemComp-SO4: |
| Source |
|
 Keywords Keywords | PROTEIN FIBRIL / protein filament / lipoprotein / glycosylation / royal jelly / major royal jelly protein / honeybee |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers






