+検索条件
-Structure paper
| タイトル | Mechanisms of activation and desensitization of full-length glycine receptor in lipid nanodiscs. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 11, Issue 1, Page 3752, Year 2020 |
| 掲載日 | 2020年7月27日 |
 著者 著者 | Arvind Kumar / Sandip Basak / Shanlin Rao / Yvonne Gicheru / Megan L Mayer / Mark S P Sansom / Sudha Chakrapani /   |
| PubMed 要旨 | Glycinergic synapses play a central role in motor control and pain processing in the central nervous system. Glycine receptors (GlyRs) are key players in mediating fast inhibitory neurotransmission ...Glycinergic synapses play a central role in motor control and pain processing in the central nervous system. Glycine receptors (GlyRs) are key players in mediating fast inhibitory neurotransmission at these synapses. While previous high-resolution structures have provided insights into the molecular architecture of GlyR, several mechanistic questions pertaining to channel function are still unanswered. Here, we present Cryo-EM structures of the full-length GlyR protein complex reconstituted into lipid nanodiscs that are captured in the unliganded (closed), glycine-bound (open and desensitized), and allosteric modulator-bound conformations. A comparison of these states reveals global conformational changes underlying GlyR channel gating and modulation. The functional state assignments were validated by molecular dynamics simulations, and the observed permeation events are in agreement with the anion selectivity and conductance of GlyR. These studies provide the structural basis for gating, ion selectivity, and single-channel conductance properties of GlyR in a lipid environment. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:32719334 / PubMed:32719334 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.07 - 3.55 Å |
| 構造データ | EMDB-20714, PDB-6ubs: EMDB-20715, PDB-6ubt: EMDB-20731, PDB-6ud3: EMDB-21234, PDB-6vm0: EMDB-21236, PDB-6vm2: EMDB-21237, PDB-6vm3: |
| 化合物 | 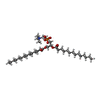 ChemComp-PX4:  ChemComp-PIO:  ChemComp-GLY:  ChemComp-NAG:  ChemComp-RI5: 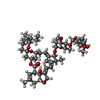 ChemComp-IVM: |
| 由来 |
|
 キーワード キーワード | MEMBRANE PROTEIN / Ions Ligands Receptors / Glycine receptor Recombinant Proteins Glycine |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について
















