+検索条件
-Structure paper
| タイトル | Architecture of the human PI4KIIIα lipid kinase complex. |
|---|---|
| ジャーナル・号・ページ | Proc Natl Acad Sci U S A, Vol. 114, Issue 52, Page 13720-13725, Year 2017 |
| 掲載日 | 2017年12月26日 |
 著者 著者 | Joshua A Lees / Yixiao Zhang / Michael S Oh / Curtis M Schauder / Xiaoling Yu / Jeremy M Baskin / Kerry Dobbs / Luigi D Notarangelo / Pietro De Camilli / Thomas Walz / Karin M Reinisch /  |
| PubMed 要旨 | Plasma membrane (PM) phosphoinositides play essential roles in cell physiology, serving as both markers of membrane identity and signaling molecules central to the cell's interaction with its ...Plasma membrane (PM) phosphoinositides play essential roles in cell physiology, serving as both markers of membrane identity and signaling molecules central to the cell's interaction with its environment. The first step in PM phosphoinositide synthesis is the conversion of phosphatidylinositol (PI) to PI4P, the precursor of PI(4,5)P and PI(3,4,5)P This conversion is catalyzed by the PI4KIIIα complex, comprising a lipid kinase, PI4KIIIα, and two regulatory subunits, TTC7 and FAM126. We here report the structure of this complex at 3.6-Å resolution, determined by cryo-electron microscopy. The proteins form an obligate ∼700-kDa superassembly with a broad surface suitable for membrane interaction, toward which the kinase active sites are oriented. The structural complexity of the assembly highlights PI4P synthesis as a major regulatory junction in PM phosphoinositide homeostasis. Our studies provide a framework for further exploring the mechanisms underlying PM phosphoinositide regulation. |
 リンク リンク |  Proc Natl Acad Sci U S A / Proc Natl Acad Sci U S A /  PubMed:29229838 / PubMed:29229838 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.6 Å |
| 構造データ | |
| 化合物 | 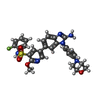 ChemComp-E4S: |
| 由来 |
|
 キーワード キーワード | Transferase/Signaling Protein /  kinase (キナーゼ) / phosphoinositide synthesis / Transferase-Signaling Protein complex kinase (キナーゼ) / phosphoinositide synthesis / Transferase-Signaling Protein complex |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について






