+検索条件
-Structure paper
| タイトル | Structural and Catalytic Insight into the Unique Pentacyclic Triterpene Synthase TwOSC. |
|---|---|
| ジャーナル・号・ページ | Angew Chem Int Ed Engl, Vol. 62, Issue 48, Page e202313429, Year 2023 |
| 掲載日 | 2023年11月27日 |
 著者 著者 | Yunfeng Luo / Xiaoli Ma / Yufan Qiu / Yun Lu / Siyu Shen / Yang Li / Haiyun Gao / Kang Chen / Jiawei Zhou / Tianyuan Hu / Lichan Tu / Huan Zhao / Dan Li / Faqiang Leng / Wei Gao / Tao Jiang / Changli Liu / Luqi Huang / Ruibo Wu / Yuru Tong /  |
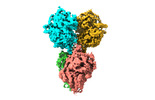
| PubMed 要旨 | The oxidosqualene cyclase (OSC) catalyzed cyclization of the linear substrate (3S)-2,3-oxidosqualene to form diverse pentacyclic triterpenoid (PT) skeletons is one of the most complex reactions in ...The oxidosqualene cyclase (OSC) catalyzed cyclization of the linear substrate (3S)-2,3-oxidosqualene to form diverse pentacyclic triterpenoid (PT) skeletons is one of the most complex reactions in nature. Friedelin has a unique PT skeleton involving a fascinating nine-step cation shuttle run (CSR) cascade rearrangement reaction, in which the carbocation formed at C2 moves to the other side of the skeleton, runs back to C3 to yield a friedelin cation, which is finally deprotonated. However, as crystal structure data of plant OSCs are lacking, it remains unknown why the CSR cascade reactions occur in friedelin biosynthesis, as does the exact catalytic mechanism of the CSR. In this study, we determined the first cryogenic electron microscopy structure of a plant OSC, friedelin synthase, from Tripterygium wilfordii Hook. f (TwOSC). We also performed quantum mechanics/molecular mechanics simulations to reveal the energy profile for the CSR cascade reaction and identify key residues crucial for PT skeleton formation. Furthermore, we semirationally designed two TwOSC mutants, which significantly improved the yields of friedelin and β-amyrin, respectively. |
 リンク リンク |  Angew Chem Int Ed Engl / Angew Chem Int Ed Engl /  PubMed:37840440 PubMed:37840440 |
| 手法 | EM (単粒子) |
| 解像度 | 4.75 Å |
| 構造データ | EMDB-35996, PDB-8j5z: |
| 化合物 |  ChemComp-BOG: |
| 由来 |
|
 キーワード キーワード | PLANT PROTEIN / oxidosqualene cyclase / Tripterygium wilfordii Hook. f. / TwOSC1 / friedelin / cryo-EM structure |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について




 tripterygium wilfordii (植物)
tripterygium wilfordii (植物)