+検索条件
-Structure paper
| タイトル | Human IFT-A complex structures provide molecular insights into ciliary transport. |
|---|---|
| ジャーナル・号・ページ | Cell Res, Vol. 33, Issue 4, Page 288-298, Year 2023 |
| 掲載日 | 2023年2月13日 |
 著者 著者 | Meiqin Jiang / Vivek Reddy Palicharla / Darcie Miller / Sun-Hee Hwang / Hanwen Zhu / Patricia Hixson / Saikat Mukhopadhyay / Ji Sun /  |
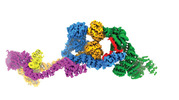
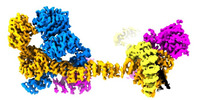
| PubMed 要旨 | Intraflagellar transport (IFT) complexes, IFT-A and IFT-B, form bidirectional trains that move along the axonemal microtubules and are essential for assembling and maintaining cilia. Mutations in IFT ...Intraflagellar transport (IFT) complexes, IFT-A and IFT-B, form bidirectional trains that move along the axonemal microtubules and are essential for assembling and maintaining cilia. Mutations in IFT subunits lead to numerous ciliopathies involving multiple tissues. However, how IFT complexes assemble and mediate cargo transport lacks mechanistic understanding due to missing high-resolution structural information of the holo-complexes. Here we report cryo-EM structures of human IFT-A complexes in the presence and absence of TULP3 at overall resolutions of 3.0-3.9 Å. IFT-A adopts a "lariat" shape with interconnected core and peripheral subunits linked by structurally vital zinc-binding domains. TULP3, the cargo adapter, interacts with IFT-A through its N-terminal region, and interface mutations disrupt cargo transport. We also determine the molecular impacts of disease mutations on complex formation and ciliary transport. Our work reveals IFT-A architecture, sheds light on ciliary transport and IFT train formation, and enables the rationalization of disease mutations in ciliopathies. |
 リンク リンク |  Cell Res / Cell Res /  PubMed:36775821 / PubMed:36775821 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.7 - 4.3 Å |
| 構造データ | EMDB-29073, PDB-8fgw: EMDB-29078, PDB-8fh3: |
| 化合物 |  ChemComp-ZN: |
| 由来 |
|
 キーワード キーワード | TRANSPORT PROTEIN / IFT-A complex / TULP3 / cilia |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について





 homo sapiens (ヒト)
homo sapiens (ヒト)