+検索条件
-Structure paper
| タイトル | Compact IF2 allows initiator tRNA accommodation into the P site and gates the ribosome to elongation. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 13, Issue 1, Page 3388, Year 2022 |
| 掲載日 | 2022年6月13日 |
 著者 著者 | Ritwika S Basu / Michael B Sherman / Matthieu G Gagnon /  |
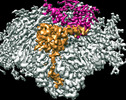
| PubMed 要旨 | During translation initiation, initiation factor 2 (IF2) holds initiator transfer RNA (fMet-tRNA) in a specific orientation in the peptidyl (P) site of the ribosome. Upon subunit joining IF2 ...During translation initiation, initiation factor 2 (IF2) holds initiator transfer RNA (fMet-tRNA) in a specific orientation in the peptidyl (P) site of the ribosome. Upon subunit joining IF2 hydrolyzes GTP and, concomitant with inorganic phosphate (P) release, changes conformation facilitating fMet-tRNA accommodation into the P site and transition of the 70 S ribosome initiation complex (70S-IC) to an elongation-competent ribosome. The mechanism by which IF2 separates from initiator tRNA at the end of translation initiation remains elusive. Here, we report cryo-electron microscopy (cryo-EM) structures of the 70S-IC from Pseudomonas aeruginosa bound to compact IF2-GDP and initiator tRNA. Relative to GTP-bound IF2, rotation of the switch 2 α-helix in the G-domain bound to GDP unlocks a cascade of large-domain movements in IF2 that propagate to the distal tRNA-binding domain C2. The C2-domain relocates 35 angstroms away from tRNA, explaining how IF2 makes way for fMet-tRNA accommodation into the P site. Our findings provide the basis by which IF2 gates the ribosome to the elongation phase. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:35697706 / PubMed:35697706 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 2.8 Å |
| 構造データ | EMDB-26553, PDB-7uiu: |
| 由来 |
|
 キーワード キーワード | RIBOSOME / Initiation Factor 2 / 70S ribosome / cryo-EM / translation initiation / initiator tRNA / conformational changes |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について




 pseudomonas aeruginosa pao1 (緑膿菌)
pseudomonas aeruginosa pao1 (緑膿菌)