+Search query
-Structure paper
| Title | Structural evidence for the partially oxidized dipyrromethene and dipyrromethanone forms of the cofactor of porphobilinogen deaminase: structures of the Bacillus megaterium enzyme at near-atomic resolution. |
|---|---|
| Journal, issue, pages | Acta Crystallogr. ,Sect. D, Vol. 70, Page 744-751, Year 2014 |
| Publish date | Sep 6, 2013 (structure data deposition date) |
 Authors Authors | Azim, N. / Deery, E. / Warren, M.J. / Wolfenden, B.A. / Erskine, P. / Cooper, J.B. / Coker, A. / Wood, S.P. / Akhtar, M. |
 External links External links |  Acta Crystallogr. ,Sect. D / Acta Crystallogr. ,Sect. D /  PubMed:24598743 PubMed:24598743 |
| Methods | X-ray diffraction |
| Resolution | 1.455 - 1.6 Å |
| Structure data |  PDB-4mlq:  PDB-4mlv: |
| Chemicals |  ChemComp-DPM: 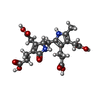 ChemComp-29P:  ChemComp-ACY:  ChemComp-HOH: |
| Source |
|
 Keywords Keywords | TRANSFERASE / tetrapyrrole biosynthesis / porphobilinogen deaminase / dipyrromethane cofactor / three-domain fold / domains 1 and 2 resemble the fold of type II periplasmic binding proteins / pyrrole polymerisation |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers



 bacillus megaterium (bacteria)
bacillus megaterium (bacteria)