+Search query
-Structure paper
| Title | The A128T Resistance Mutation Reveals Aberrant Protein Multimerization as the Primary Mechanism of Action of Allosteric HIV-1 Integrase Inhibitors. |
|---|---|
| Journal, issue, pages | J. Biol. Chem., Vol. 288, Page 15813-15820, Year 2013 |
| Publish date | Aug 30, 2012 (structure data deposition date) |
 Authors Authors | Feng, L. / Sharma, A. / Slaughter, A. / Jena, N. / Koh, Y. / Shkriabai, N. / Larue, R.C. / Patel, P.A. / Mitsuya, H. / Kessl, J.J. ...Feng, L. / Sharma, A. / Slaughter, A. / Jena, N. / Koh, Y. / Shkriabai, N. / Larue, R.C. / Patel, P.A. / Mitsuya, H. / Kessl, J.J. / Engelman, A. / Fuchs, J.R. / Kvaratskhelia, M. |
 External links External links |  J. Biol. Chem. / J. Biol. Chem. /  PubMed:23615903 PubMed:23615903 |
| Methods | X-ray diffraction |
| Resolution | 2.09 - 2.65 Å |
| Structure data |  PDB-4gvm:  PDB-4gw6:  PDB-4jlh: |
| Chemicals |  ChemComp-ARS: 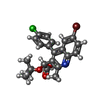 ChemComp-LF2:  ChemComp-HOH: 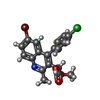 ChemComp-0L9:  ChemComp-SO4: |
| Source |
|
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR / Integrase / CCD / DDE motif / dimer interface / allosteric inhibitor / drug resistance / HYDROLASE-HYDROLASE INHIBITOR complex / TRANSFERASE/TRANSFERASE INHIBITOR / A128T mutation / TRANSFERASE-TRANSFERASE INHIBITOR complex |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers



 human immunodeficiency virus type 1 (new york-5 isolate)
human immunodeficiency virus type 1 (new york-5 isolate) human immunodeficiency virus type 1
human immunodeficiency virus type 1