+Search query
-Structure paper
| Title | Broadly neutralizing antibodies target a haemagglutinin anchor epitope. |
|---|---|
| Journal, issue, pages | Nature, Vol. 602, Issue 7896, Page 314-320, Year 2022 |
| Publish date | Dec 23, 2021 |
 Authors Authors | Jenna J Guthmiller / Julianna Han / Henry A Utset / Lei Li / Linda Yu-Ling Lan / Carole Henry / Christopher T Stamper / Meagan McMahon / George O'Dell / Monica L Fernández-Quintero / Alec W Freyn / Fatima Amanat / Olivia Stovicek / Lauren Gentles / Sara T Richey / Alba Torrents de la Peña / Victoria Rosado / Haley L Dugan / Nai-Ying Zheng / Micah E Tepora / Dalia J Bitar / Siriruk Changrob / Shirin Strohmeier / Min Huang / Adolfo García-Sastre / Klaus R Liedl / Jesse D Bloom / Raffael Nachbagauer / Peter Palese / Florian Krammer / Lynda Coughlan / Andrew B Ward / Patrick C Wilson /   |
| PubMed Abstract | Broadly neutralizing antibodies that target epitopes of haemagglutinin on the influenza virus have the potential to provide near universal protection against influenza virus infection. However, viral ...Broadly neutralizing antibodies that target epitopes of haemagglutinin on the influenza virus have the potential to provide near universal protection against influenza virus infection. However, viral mutants that escape broadly neutralizing antibodies have been reported. The identification of broadly neutralizing antibody classes that can neutralize viral escape mutants is critical for universal influenza virus vaccine design. Here we report a distinct class of broadly neutralizing antibodies that target a discrete membrane-proximal anchor epitope of the haemagglutinin stalk domain. Anchor epitope-targeting antibodies are broadly neutralizing across H1 viruses and can cross-react with H2 and H5 viruses that are a pandemic threat. Antibodies that target this anchor epitope utilize a highly restricted repertoire, which encodes two public binding motifs that make extensive contacts with conserved residues in the fusion peptide. Moreover, anchor epitope-targeting B cells are common in the human memory B cell repertoire and were recalled in humans by an oil-in-water adjuvanted chimeric haemagglutinin vaccine, which is a potential universal influenza virus vaccine. To maximize protection against seasonal and pandemic influenza viruses, vaccines should aim to boost this previously untapped source of broadly neutralizing antibodies that are widespread in the human memory B cell pool. |
 External links External links |  Nature / Nature /  PubMed:34942633 / PubMed:34942633 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 3.38 - 20.0 Å |
| Structure data |  EMDB-25634:  EMDB-25635:  EMDB-25636:  EMDB-25637:  EMDB-25638: 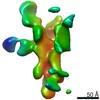 EMDB-25639:  EMDB-25640:  EMDB-25641: 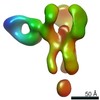 EMDB-25642: 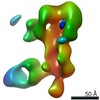 EMDB-25643: 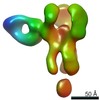 EMDB-25644:  EMDB-25645:  EMDB-25646: EMDB-25655, PDB-7t3d: |
| Chemicals |  ChemComp-NAG: |
| Source |
|
 Keywords Keywords | Viral Protein/IMMUNE SYSTEM / anchor / antibodies / influenza A virus / hemagglutinin / IMMUNE SYSTEM / Viral Protein-IMMUNE SYSTEM complex |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers





 homo sapiens (human)
homo sapiens (human) influenza a virus (a/california/04/2009(h1n1))
influenza a virus (a/california/04/2009(h1n1))