+Search query
-Structure paper
| Title | Structural basis of menaquinone reduction by succinate dehydrogenase from Chloroflexus aurantiacus. |
|---|---|
| Journal, issue, pages | Nat Commun, Vol. 16, Issue 1, Page 10782, Year 2025 |
| Publish date | Nov 28, 2025 |
 Authors Authors | Xin Zhang / Jingyi Wu / Jiamao Wang / Huimin He / Aokun Liu / Xin Hong / Yuanyi Yu / Xinkai Pei / Xianjie Fang / Yueyong Xin / Lu Yu / Changlin Tian / Xiaoling Xu /  |
| PubMed Abstract | Succinate: menaquinone oxidoreductase (SQR) couples the oxidation of succinate with the reduction of menaquinone (MK) as part of the TCA cycle and the aerobic respiratory chain in MK-containing ...Succinate: menaquinone oxidoreductase (SQR) couples the oxidation of succinate with the reduction of menaquinone (MK) as part of the TCA cycle and the aerobic respiratory chain in MK-containing bacteria and archaea. Despite its significance, questions persist regarding the electron and proton transfer mechanisms that drive the endergonic MK reduction by succinate. In this study, we determine cryo-EM structures of succinate dehydrogenase (SDH) from Chloroflexus aurantiacus (CaSDH), a facultative filamentous anoxygenic phototroph (FAP) that forms one of the earliest branches of photosynthetic bacteria. The structures of trimeric CaSDH, resolved in both apo- and MK-bound forms, reveal a single membrane-anchoring subunit containing two b-type hemes, a canonical Q site, and a Q site with atypical location, configuration and specificity, each bound to MK molecules. Using structural analysis, EPR, and enzymatic assays, we uncover electron transfer pathways connecting succinate oxidation to MK reduction at the Q and Q sites. These findings provide structural insights into the electron and proton transfer mechanisms of MK-dependent diheme SQRs and establish a foundation for structure-based inhibitor design and antibacterial drug development targeting these enzymes. |
 External links External links |  Nat Commun / Nat Commun /  PubMed:41315263 / PubMed:41315263 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 2.62 - 2.9 Å |
| Structure data | EMDB-62932, PDB-9lay: EMDB-62933, PDB-9laz: EMDB-62934, PDB-9lb0: EMDB-62935, PDB-9lb1: |
| Chemicals |  ChemComp-FAD: 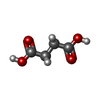 ChemComp-SIN:  ChemComp-SF4:  ChemComp-FES:  ChemComp-F3S:  ChemComp-HEM:  ChemComp-LMT:  ChemComp-PGV:  ChemComp-PEV:  ChemComp-MQ7: 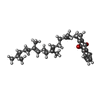 ChemComp-1L3: |
| Source |
|
 Keywords Keywords | MEMBRANE PROTEIN / Succinate: menaquinone oxidoreductase; membrane protein; complex II; electron transfer; MEMBRANE PROTEIN / succinate: menaquinone oxidoreductase; succinate dehydrogenase; electron transfer; MEMBRANE PROTEIN / Succinate: menaquinone oxidoreductase; 3-hydroxypropionate cycle; succinate dehydrogenase;electron transfer |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers












 chloroflexus aurantiacus j-10-fl (bacteria)
chloroflexus aurantiacus j-10-fl (bacteria)