+検索条件
-Structure paper
| タイトル | Structural insights into the iron nitrogenase complex. |
|---|---|
| ジャーナル・号・ページ | Nat Struct Mol Biol, Vol. 31, Issue 1, Page 150-158, Year 2024 |
| 掲載日 | 2023年12月7日 |
 著者 著者 | Frederik V Schmidt / Luca Schulz / Jan Zarzycki / Simone Prinz / Niels N Oehlmann / Tobias J Erb / Johannes G Rebelein /  |
| PubMed 要旨 | Nitrogenases are best known for catalyzing the reduction of dinitrogen to ammonia at a complex metallic cofactor. Recently, nitrogenases were shown to reduce carbon dioxide (CO) and carbon monoxide ...Nitrogenases are best known for catalyzing the reduction of dinitrogen to ammonia at a complex metallic cofactor. Recently, nitrogenases were shown to reduce carbon dioxide (CO) and carbon monoxide to hydrocarbons, offering a pathway to recycle carbon waste into hydrocarbon products. Among the three nitrogenase isozymes, the iron nitrogenase has the highest wild-type activity for the reduction of CO, but the molecular architecture facilitating these activities has remained unknown. Here, we report a 2.35-Å cryogenic electron microscopy structure of the ADP·AlF-stabilized iron nitrogenase complex from Rhodobacter capsulatus, revealing an [FeSC-(R)-homocitrate] cluster in the active site. The enzyme complex suggests that the iron nitrogenase G subunit is involved in cluster stabilization and substrate channeling and confers specificity between nitrogenase reductase and catalytic component proteins. Moreover, the structure highlights a different interface between the two catalytic halves of the iron and the molybdenum nitrogenase, potentially influencing the intrasubunit 'communication' and thus the nitrogenase mechanism. |
 リンク リンク |  Nat Struct Mol Biol / Nat Struct Mol Biol /  PubMed:38062208 / PubMed:38062208 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 2.35 - 2.49 Å |
| 構造データ | EMDB-16890, PDB-8oie: EMDB-17583, PDB-8pbb: |
| 化合物 | 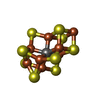 ChemComp-S5Q: 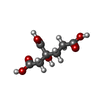 ChemComp-HCA: 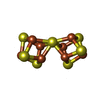 ChemComp-CLF:  ChemComp-ADP:  ChemComp-MG:  ChemComp-AF3:  ChemComp-SF4:  ChemComp-HOH: |
| 由来 |
|
 キーワード キーワード | OXIDOREDUCTASE / nitrogen fixation / Fe nitrogenase |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について







 rhodobacter capsulatus sb 1003 (バクテリア)
rhodobacter capsulatus sb 1003 (バクテリア)