+検索条件
-Structure paper
| タイトル | Mechanism of action for small-molecule inhibitors of triacylglycerol synthesis. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 14, Issue 1, Page 3100, Year 2023 |
| 掲載日 | 2023年5月29日 |
 著者 著者 | Xuewu Sui / Kun Wang / Kangkang Song / Chen Xu / Jiunn Song / Chia-Wei Lee / Maofu Liao / Robert V Farese / Tobias C Walther /   |
| PubMed 要旨 | Inhibitors of triacylglycerol (TG) synthesis have been developed to treat metabolism-related diseases, but we know little about their mechanisms of action. Here, we report cryo-EM structures of the ...Inhibitors of triacylglycerol (TG) synthesis have been developed to treat metabolism-related diseases, but we know little about their mechanisms of action. Here, we report cryo-EM structures of the TG-synthesis enzyme acyl-CoA:diacylglycerol acyltransferase 1 (DGAT1), a membrane bound O-acyltransferase (MBOAT), in complex with two different inhibitors, T863 and DGAT1IN1. Each inhibitor binds DGAT1's fatty acyl-CoA substrate binding tunnel that opens to the cytoplasmic side of the ER. T863 blocks access to the tunnel entrance, whereas DGAT1IN1 extends further into the enzyme, with an amide group interacting with more deeply buried catalytic residues. A survey of DGAT1 inhibitors revealed that this amide group may serve as a common pharmacophore for inhibition of MBOATs. The inhibitors were minimally active against the related MBOAT acyl-CoA:cholesterol acyltransferase 1 (ACAT1), yet a single-residue mutation sensitized ACAT1 for inhibition. Collectively, our studies provide a structural foundation for developing DGAT1 and other MBOAT inhibitors. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:37248213 / PubMed:37248213 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.2 Å |
| 構造データ | EMDB-28577, PDB-8esm: EMDB-28594, PDB-8etm: |
| 化合物 |  ChemComp-WS0: 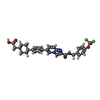 ChemComp-WTT: |
| 由来 |
|
 キーワード キーワード | TRANSFERASE/TRANSFERASE INHIBITOR / triglyceride biosynthesis / lipid storage / lipid metabolism / membrane protein / TRANSFERASE-TRANSFERASE INHIBITOR complex |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について







 homo sapiens (ヒト)
homo sapiens (ヒト)