+検索条件
-Structure paper
| タイトル | Open-state structure and pore gating mechanism of the cardiac sodium channel. |
|---|---|
| ジャーナル・号・ページ | Cell, Vol. 184, Issue 20, Page 5151-5162.e11, Year 2021 |
| 掲載日 | 2021年9月30日 |
 著者 著者 | Daohua Jiang / Richard Banh / Tamer M Gamal El-Din / Lige Tonggu / Michael J Lenaeus / Régis Pomès / Ning Zheng / William A Catterall /    |
| PubMed 要旨 | The heartbeat is initiated by voltage-gated sodium channel Na1.5, which opens rapidly and triggers the cardiac action potential; however, the structural basis for pore opening remains unknown. Here, ...The heartbeat is initiated by voltage-gated sodium channel Na1.5, which opens rapidly and triggers the cardiac action potential; however, the structural basis for pore opening remains unknown. Here, we blocked fast inactivation with a mutation and captured the elusive open-state structure. The fast inactivation gate moves away from its receptor, allowing asymmetric opening of pore-lining S6 segments, which bend and rotate at their intracellular ends to dilate the activation gate to ∼10 Å diameter. Molecular dynamics analyses predict physiological rates of Na conductance. The open-state pore blocker propafenone binds in a high-affinity pose, and drug-access pathways are revealed through the open activation gate and fenestrations. Comparison with mutagenesis results provides a structural map of arrhythmia mutations that target the activation and fast inactivation gates. These results give atomic-level insights into molecular events that underlie generation of the action potential, open-state drug block, and fast inactivation of cardiac sodium channels, which initiate the heartbeat. |
 リンク リンク |  Cell / Cell /  PubMed:34520724 / PubMed:34520724 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.4 Å |
| 構造データ | EMDB-31519, PDB-7fbs: |
| 化合物 |  ChemComp-NAG:  ChemComp-6OU: 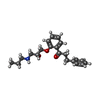 ChemComp-4Y4: 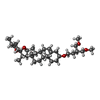 ChemComp-9Z9: |
| 由来 |
|
 キーワード キーワード | MEMBRANE PROTEIN / ion channel / four-domain |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について






 recombinant vesicular stomatitis indiana virus rvsv-g/gfp (ウイルス)
recombinant vesicular stomatitis indiana virus rvsv-g/gfp (ウイルス)