+検索条件
-Structure paper
| タイトル | Architecture of human mTOR complex 1. |
|---|---|
| ジャーナル・号・ページ | Science, Vol. 351, Issue 6268, Page 48-52, Year 2016 |
| 掲載日 | 2016年1月1日 |
 著者 著者 | Christopher H S Aylett / Evelyn Sauer / Stefan Imseng / Daniel Boehringer / Michael N Hall / Nenad Ban / Timm Maier /  |
| PubMed 要旨 | Target of rapamycin (TOR), a conserved protein kinase and central controller of cell growth, functions in two structurally and functionally distinct complexes: TORC1 and TORC2. Dysregulation of ...Target of rapamycin (TOR), a conserved protein kinase and central controller of cell growth, functions in two structurally and functionally distinct complexes: TORC1 and TORC2. Dysregulation of mammalian TOR (mTOR) signaling is implicated in pathologies that include diabetes, cancer, and neurodegeneration. We resolved the architecture of human mTORC1 (mTOR with subunits Raptor and mLST8) bound to FK506 binding protein (FKBP)-rapamycin, by combining cryo-electron microscopy at 5.9 angstrom resolution with crystallographic studies of Chaetomium thermophilum Raptor at 4.3 angstrom resolution. The structure explains how FKBP-rapamycin and architectural elements of mTORC1 limit access to the recessed active site. Consistent with a role in substrate recognition and delivery, the conserved amino-terminal domain of Raptor is juxtaposed to the kinase active site. |
 リンク リンク |  Science / Science /  PubMed:26678875 PubMed:26678875 |
| 手法 | EM (単粒子) / X線回折 |
| 解像度 | 4.3 - 6.1 Å |
| 構造データ |  EMDB-3212: EMDB-3213, PDB-5flc:  PDB-5ef5: |
| 化合物 | 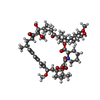 ChemComp-RAP: |
| 由来 |
|
 キーワード キーワード | SIGNALING PROTEIN / mTORC1 / Target of Rapamycin / Raptor / Rptor / TRANSFERASE / RAPAMYCIN |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について





 homo sapiens (ヒト)
homo sapiens (ヒト) chaetomium thermophilum (菌類)
chaetomium thermophilum (菌類)
