+Search query
-Structure paper
| Title | The crystal structure of C176A mutated [Fe]-hydrogenase suggests an acyl-iron ligation in the active site iron complex. |
|---|---|
| Journal, issue, pages | Febs Lett., Vol. 583, Page 585-590, Year 2009 |
| Publish date | Oct 31, 2008 (structure data deposition date) |
 Authors Authors | Hiromoto, T. / Ataka, K. / Pilak, O. / Vogt, S. / Stagni, M.S. / Meyer-Klaucke, W. / Warkentin, E. / Thauer, R.K. / Shima, S. / Ermler, U. |
 External links External links |  Febs Lett. / Febs Lett. /  PubMed:19162018 PubMed:19162018 |
| Methods | X-ray diffraction |
| Resolution | 1.75 - 1.95 Å |
| Structure data |  PDB-3f46:  PDB-3f47: |
| Chemicals | 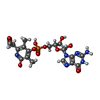 ChemComp-I2C:  ChemComp-FE2: 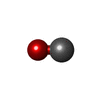 ChemComp-CMO:  ChemComp-DTV:  ChemComp-HOH:  ChemComp-NA: |
| Source |
|
 Keywords Keywords | OXIDOREDUCTASE / ROSSMANN FOLD / HELIX BUNDLE / COMPLEX WITH IRON GUANYLYL PYRIDINOL COFACTOR / C176A MUTANT / Methanogenesis / One-carbon metabolism |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers




 methanocaldococcus jannaschii (archaea)
methanocaldococcus jannaschii (archaea)