[English] 日本語
 Yorodumi
Yorodumi- EMDB-41194: Human cytomegalovirus portal vertex, virion configuration 1 (VC1) -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human cytomegalovirus portal vertex, virion configuration 1 (VC1) | ||||||||||||||||||||||||||||||
 Map data Map data | Post-processed map used for real space refinement. | ||||||||||||||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||||||||||||||
 Keywords Keywords | tegument / portal / DNA packaging / intracellular transport / VIRUS | ||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationmicrotubule-dependent intracellular transport of viral material towards cell periphery / host cell viral assembly compartment / T=16 icosahedral viral capsid / viral tegument / viral DNA genome packaging / viral capsid assembly / host cell cytoplasmic vesicle / virion assembly / chromosome organization / viral process ...microtubule-dependent intracellular transport of viral material towards cell periphery / host cell viral assembly compartment / T=16 icosahedral viral capsid / viral tegument / viral DNA genome packaging / viral capsid assembly / host cell cytoplasmic vesicle / virion assembly / chromosome organization / viral process / viral penetration into host nucleus / viral capsid / host cell / host cell Golgi apparatus / symbiont-mediated perturbation of host ubiquitin-like protein modification / ubiquitinyl hydrolase 1 / host cell cytoplasm / cysteine-type deubiquitinase activity / Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases / host cell perinuclear region of cytoplasm / symbiont entry into host cell / host cell nucleus / structural molecule activity / proteolysis / DNA binding / identical protein binding Similarity search - Function | ||||||||||||||||||||||||||||||
| Biological species |   Human herpesvirus 5 strain AD169 Human herpesvirus 5 strain AD169 | ||||||||||||||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.5 Å | ||||||||||||||||||||||||||||||
 Authors Authors | Jih J / Liu YT / Liu W / Zhou H | ||||||||||||||||||||||||||||||
| Funding support |  United States, 9 items United States, 9 items
| ||||||||||||||||||||||||||||||
 Citation Citation |  Journal: Sci Adv / Year: 2024 Journal: Sci Adv / Year: 2024Title: The incredible bulk: Human cytomegalovirus tegument architectures uncovered by AI-empowered cryo-EM. Authors: Jonathan Jih / Yun-Tao Liu / Wei Liu / Z Hong Zhou /  Abstract: The compartmentalization of eukaryotic cells presents considerable challenges to the herpesvirus life cycle. The herpesvirus tegument, a bulky proteinaceous aggregate sandwiched between ...The compartmentalization of eukaryotic cells presents considerable challenges to the herpesvirus life cycle. The herpesvirus tegument, a bulky proteinaceous aggregate sandwiched between herpesviruses' capsid and envelope, is uniquely evolved to address these challenges, yet tegument structure and organization remain poorly characterized. We use deep-learning-enhanced cryogenic electron microscopy to investigate the tegument of human cytomegalovirus virions and noninfectious enveloped particles (NIEPs; a genome packaging-aborted state), revealing a portal-biased tegumentation scheme. We resolve atomic structures of portal vertex-associated tegument (PVAT) and identify multiple configurations of PVAT arising from layered reorganization of pUL77, pUL48 (large tegument protein), and pUL47 (inner tegument protein) assemblies. Analyses show that pUL77 seals the last-packaged viral genome end through electrostatic interactions, pUL77 and pUL48 harbor a head-linker-capsid-binding motif conducive to PVAT reconfiguration, and pUL47/48 dimers form 45-nm-long filaments extending from the portal vertex. These results provide a structural framework for understanding how herpesvirus tegument facilitates and evolves during processes spanning viral genome packaging to delivery. | ||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41194.map.gz emd_41194.map.gz | 166.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41194-v30.xml emd-41194-v30.xml emd-41194.xml emd-41194.xml | 33.4 KB 33.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_41194_fsc.xml emd_41194_fsc.xml | 12.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_41194.png emd_41194.png | 214.8 KB | ||
| Filedesc metadata |  emd-41194.cif.gz emd-41194.cif.gz | 11 KB | ||
| Others |  emd_41194_additional_1.map.gz emd_41194_additional_1.map.gz emd_41194_half_map_1.map.gz emd_41194_half_map_1.map.gz emd_41194_half_map_2.map.gz emd_41194_half_map_2.map.gz | 140.1 MB 140.9 MB 141 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41194 http://ftp.pdbj.org/pub/emdb/structures/EMD-41194 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41194 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41194 | HTTPS FTP |
-Related structure data
| Related structure data |  8tepMC  8tesC  8tetC  8teuC  8tewC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_41194.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41194.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Post-processed map used for real space refinement. | ||||||||||||||||||||
| Voxel size | X=Y=Z: 1.36 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Primary map.
| File | emd_41194_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Primary map. | ||||||||||||
| Projections & Slices |
| ||||||||||||
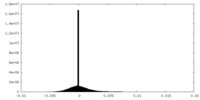
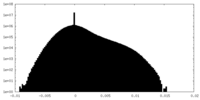
| Density Histograms |
-Half map: Half map 1.
| File | emd_41194_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map 2.
| File | emd_41194_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Human herpesvirus 5 strain AD169
+Supramolecule #1: Human herpesvirus 5 strain AD169
+Macromolecule #1: Large tegument protein deneddylase
+Macromolecule #2: Inner tegument protein
+Macromolecule #3: Capsid vertex component 2
+Macromolecule #4: Capsid vertex component 1
+Macromolecule #5: Major capsid protein
+Macromolecule #6: Small capsomere-interacting protein
+Macromolecule #7: Triplex capsid protein 1
+Macromolecule #8: Triplex capsid protein 2
+Macromolecule #9: Large structural phosphoprotein
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Grid | Model: Quantifoil R2/1 / Material: COPPER / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Average exposure time: 8.0 sec. / Average electron dose: 47.2 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller













 Z
Z Y
Y X
X



















 Homo sapiens (human)
Homo sapiens (human)
