+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human VPS35L/VPS29/VPS26C Complex | |||||||||
 Map data Map data | Retriever VPS35L/VPS29/VPS26C Complex | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | COMMD / Retriever / Commander / CCC / PROTEIN TRANSPORT | |||||||||
| Function / homology |  Function and homology information Function and homology informationretromer, cargo-selective complex / WNT ligand biogenesis and trafficking / retromer complex / Golgi to plasma membrane transport / endocytic recycling / retrograde transport, endosome to Golgi / ficolin-1-rich granule membrane / intracellular protein transport / late endosome / protein transport ...retromer, cargo-selective complex / WNT ligand biogenesis and trafficking / retromer complex / Golgi to plasma membrane transport / endocytic recycling / retrograde transport, endosome to Golgi / ficolin-1-rich granule membrane / intracellular protein transport / late endosome / protein transport / early endosome / endosome / endosome membrane / Neutrophil degranulation / metal ion binding / nucleus / plasma membrane / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
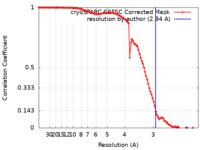
| Method | single particle reconstruction / cryo EM / Resolution: 2.94 Å | |||||||||
 Authors Authors | Chen Z / Chen B / Burstein E / Han Y | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2024 Journal: Nat Struct Mol Biol / Year: 2024Title: Structural organization of the retriever-CCC endosomal recycling complex. Authors: Daniel J Boesch / Amika Singla / Yan Han / Daniel A Kramer / Qi Liu / Kohei Suzuki / Puneet Juneja / Xuefeng Zhao / Xin Long / Michael J Medlyn / Daniel D Billadeau / Zhe Chen / Baoyu Chen / Ezra Burstein /  Abstract: The recycling of membrane proteins from endosomes to the cell surface is vital for cell signaling and survival. Retriever, a trimeric complex of vacuolar protein-sorting-associated protein (VPS)35L, ...The recycling of membrane proteins from endosomes to the cell surface is vital for cell signaling and survival. Retriever, a trimeric complex of vacuolar protein-sorting-associated protein (VPS)35L, VPS26C and VPS29, together with the CCC complex comprising coiled-coil domain-containing (CCDC)22, CCDC93 and copper metabolism domain-containing (COMMD) proteins, plays a crucial role in this process. The precise mechanisms underlying retriever assembly and its interaction with CCC have remained elusive. Here, we present a high-resolution structure of retriever in humans determined using cryogenic electron microscopy. The structure reveals a unique assembly mechanism, distinguishing it from its remotely related paralog retromer. By combining AlphaFold predictions and biochemical, cellular and proteomic analyses, we further elucidate the structural organization of the entire retriever-CCC complex across evolution and uncover how cancer-associated mutations in humans disrupt complex formation and impair membrane protein homeostasis. These findings provide a fundamental framework for understanding the biological and pathological implications associated with retriever-CCC-mediated endosomal recycling. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_40885.map.gz emd_40885.map.gz | 91.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-40885-v30.xml emd-40885-v30.xml emd-40885.xml emd-40885.xml | 23 KB 23 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_40885_fsc.xml emd_40885_fsc.xml | 10.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_40885.png emd_40885.png | 58.4 KB | ||
| Masks |  emd_40885_msk_1.map emd_40885_msk_1.map | 103 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-40885.cif.gz emd-40885.cif.gz | 7.3 KB | ||
| Others |  emd_40885_half_map_1.map.gz emd_40885_half_map_1.map.gz emd_40885_half_map_2.map.gz emd_40885_half_map_2.map.gz | 95.5 MB 95.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-40885 http://ftp.pdbj.org/pub/emdb/structures/EMD-40885 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40885 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40885 | HTTPS FTP |
-Related structure data
| Related structure data |  8synMC  8symC  8syoC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_40885.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_40885.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Retriever VPS35L/VPS29/VPS26C Complex | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.0624 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_40885_msk_1.map emd_40885_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half Map 1
| File | emd_40885_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half Map 2
| File | emd_40885_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Trimeric Complex of full-length VPS35L/VPS29/VPS26C
| Entire | Name: Trimeric Complex of full-length VPS35L/VPS29/VPS26C |
|---|---|
| Components |
|
-Supramolecule #1: Trimeric Complex of full-length VPS35L/VPS29/VPS26C
| Supramolecule | Name: Trimeric Complex of full-length VPS35L/VPS29/VPS26C / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: VPS35 endosomal protein-sorting factor-like
| Macromolecule | Name: VPS35 endosomal protein-sorting factor-like / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 109.700453 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MAVFPWHSRN RNYKAEFASC RLEAVPLEFG DYHPLKPITV TESKTKKVNR KGSTSSTSSS SSSSVVDPLS SVLDGTDPLS MFAATADPA ALAAAMDSSR RKRDRDDNSV VGSDFEPWTN KRGEILARYT TTEKLSINLF MGSEKGKAGT ATLAMSEKVR T RLEELDDF ...String: MAVFPWHSRN RNYKAEFASC RLEAVPLEFG DYHPLKPITV TESKTKKVNR KGSTSSTSSS SSSSVVDPLS SVLDGTDPLS MFAATADPA ALAAAMDSSR RKRDRDDNSV VGSDFEPWTN KRGEILARYT TTEKLSINLF MGSEKGKAGT ATLAMSEKVR T RLEELDDF EEGSQKELLN LTQQDYVNRI EELNQSLKDA WASDQKVKAL KIVIQCSKLL SDTSVIQFYP SKFVLITDIL DT FGKLVYE RIFSMCVDSR SVLPDHFSPE NANDTAKETC LNWFFKIASI RELIPRFYVE ASILKCNKFL SKTGISECLP RLT CMIRGI GDPLVSVYAR AYLCRVGMEV APHLKETLNK NFFDFLLTFK QIHGDTVQNQ LVVQGVELPS YLPLYPPAMD WIFQ CISYH APEALLTEMM ERCKKLGNNA LLLNSVMSAF RAEFIATRSM DFIGMIKECD ESGFPKHLLF RSLGLNLALA DPPES DRLQ ILNEAWKVIT KLKNPQDYIN CAEVWVEYTC KHFTKREVNT VLADVIKHMT PDRAFEDSYP QLQLIIKKVI AHFHDF SVL FSVEKFLPFL DMFQKESVRV EVCKCIMDAF IKHQQEPTKD PVILNALLHV CKTMHDSVNA LTLEDEKRML SYLINGF IK MVSFGRDFEQ QLSFYVESRS MFCNLEPVLV QLIHSVNRLA METRKVMKGN HSRKTAAFVR ACVAYCFITI PSLAGIFT R LNLYLHSGQV ALANQCLSQA DAFFKAAISL VPEVPKMINI DGKMRPSESF LLEFLCNFFS TLLIVPDHPE HGVLFLVRE LLNVIQDYTW EDNSDEKIRI YTCVLHLLSA MSQETYLYHI DKVDSNDSLY GGDSKFLAEN NKLCETVMAQ ILEHLKTLAK DEALKRQSS LGLSFFNSIL AHGDLRNNKL NQLSVNLWHL AQRHGCADTR TMVKTLEYIK KQSKQPDMTH LTELALRLPL Q TRT UniProtKB: VPS35 endosomal protein-sorting factor-like |
-Macromolecule #2: Vacuolar protein sorting-associated protein 29
| Macromolecule | Name: Vacuolar protein sorting-associated protein 29 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 23.03832 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MAGHRLVLVL GDLHIPHRCN SLPAKFKKLL VPGKIQHILC TGNLCTKESY DYLKTLAGDV HIVRGDFDEN LNYPEQKVVT VGQFKIGLI HGHQVIPWGD MASLALLQRQ FDVDILISGH THKFEAFEHE NKFYINPGSA TGAYNALETN IIPSFVLMDI Q ASTVVTYV ...String: MAGHRLVLVL GDLHIPHRCN SLPAKFKKLL VPGKIQHILC TGNLCTKESY DYLKTLAGDV HIVRGDFDEN LNYPEQKVVT VGQFKIGLI HGHQVIPWGD MASLALLQRQ FDVDILISGH THKFEAFEHE NKFYINPGSA TGAYNALETN IIPSFVLMDI Q ASTVVTYV YQLIGDDVKV ERIEYKKPEN LYFQGGGSGG SHHHHHH UniProtKB: Vacuolar protein sorting-associated protein 29 |
-Macromolecule #3: Vacuolar protein sorting-associated protein 26C
| Macromolecule | Name: Vacuolar protein sorting-associated protein 26C / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 33.049344 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGTALDIKIK RANKVYHAGE VLSGVVVISS KDSVQHQGVS LTMEGTVNLQ LSAKSVGVFE AFYNSVKPIQ IINSTIEMVK PGKFPSGKT EIPFEFPLHL KGNKVLYETY HGVFVNIQYT LRCDMKRSLL AKDLTKTCEF IVHSAPQKGK FTPSPVDFTI T PETLQNVK ...String: MGTALDIKIK RANKVYHAGE VLSGVVVISS KDSVQHQGVS LTMEGTVNLQ LSAKSVGVFE AFYNSVKPIQ IINSTIEMVK PGKFPSGKT EIPFEFPLHL KGNKVLYETY HGVFVNIQYT LRCDMKRSLL AKDLTKTCEF IVHSAPQKGK FTPSPVDFTI T PETLQNVK ERALLPKFLL RGHLNSTNCV ITQPLTGELV VESSEAAIRS VELQLVRVET CGCAEGYARD ATEIQNIQIA DG DVCRGLS VPIYMVFPRL FTCPTLETTN FKVEFEVNIV VLLHPDHLIT ENFPLKLCRI UniProtKB: Vacuolar protein sorting-associated protein 26C |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.25 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7 Component:
Details: 10 mM HEPES (pH 7.0), 150 mM NaCl, 2 mM MgCl2, 2 mM DTT, and 5% (v/v) glycerol | ||||||||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE | ||||||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 278.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Quantum ER / Energy filter - Slit width: 20 eV |
| Software | Name: SerialEM |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number grids imaged: 1 / Number real images: 3594 / Average exposure time: 5.0 sec. / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.4 µm / Nominal defocus min: 1.2 µm / Nominal magnification: 105000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | Chain - Source name: AlphaFold / Chain - Initial model type: in silico model |
|---|---|
| Software | Name:  Coot Coot |
| Refinement | Space: REAL / Overall B value: 107.9 |
| Output model |  PDB-8syn: |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)