+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cleaved Ycf1p Dimer in the IFwide-beta conformation | |||||||||
 Map data Map data | Cleaved Ycf1p Dimer in the IFwide-beta conformation | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ABC transporter / Dimer / TRANSPORT PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationABC-type Cd2+ transporter / ABC-type cadmium transporter activity / Recycling of bile acids and salts / Heme degradation / Aspirin ADME / Atorvastatin ADME / Paracetamol ADME / P-type cadmium transporter activity / bilirubin transmembrane transporter activity / bilirubin transport ...ABC-type Cd2+ transporter / ABC-type cadmium transporter activity / Recycling of bile acids and salts / Heme degradation / Aspirin ADME / Atorvastatin ADME / Paracetamol ADME / P-type cadmium transporter activity / bilirubin transmembrane transporter activity / bilirubin transport / vacuole fusion, non-autophagic / ABC-family proteins mediated transport / ABC-type glutathione-S-conjugate transporter / ABC-type glutathione S-conjugate transporter activity / fungal-type vacuole membrane / response to metal ion / ATPase-coupled transmembrane transporter activity / response to cadmium ion / glutathione metabolic process / cell redox homeostasis / transmembrane transport / membrane raft / ATP hydrolysis activity / ATP binding / membrane Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | |||||||||
 Authors Authors | Bickers SC / Benlekbir S / Rubinstein JL / Kanelis V | |||||||||
| Funding support |  Canada, 1 items Canada, 1 items
| |||||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Structure of a dimeric ABC transporter Authors: Bickers SC / Benlekbir S / Rubinstein JL / Kanelis V | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_40692.map.gz emd_40692.map.gz | 154.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-40692-v30.xml emd-40692-v30.xml emd-40692.xml emd-40692.xml | 16.4 KB 16.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_40692.png emd_40692.png | 182.9 KB | ||
| Filedesc metadata |  emd-40692.cif.gz emd-40692.cif.gz | 6.3 KB | ||
| Others |  emd_40692_half_map_1.map.gz emd_40692_half_map_1.map.gz emd_40692_half_map_2.map.gz emd_40692_half_map_2.map.gz | 151.7 MB 151.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-40692 http://ftp.pdbj.org/pub/emdb/structures/EMD-40692 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40692 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40692 | HTTPS FTP |
-Validation report
| Summary document |  emd_40692_validation.pdf.gz emd_40692_validation.pdf.gz | 880 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_40692_full_validation.pdf.gz emd_40692_full_validation.pdf.gz | 879.6 KB | Display | |
| Data in XML |  emd_40692_validation.xml.gz emd_40692_validation.xml.gz | 14.9 KB | Display | |
| Data in CIF |  emd_40692_validation.cif.gz emd_40692_validation.cif.gz | 17.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-40692 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-40692 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-40692 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-40692 | HTTPS FTP |
-Related structure data
| Related structure data |  8sq0MC  8sqmC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_40692.map.gz / Format: CCP4 / Size: 163.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_40692.map.gz / Format: CCP4 / Size: 163.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cleaved Ycf1p Dimer in the IFwide-beta conformation | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.03 Å | ||||||||||||||||||||||||||||||||||||
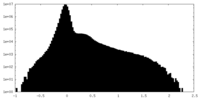
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Half Map 1
| File | emd_40692_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
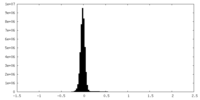
| Density Histograms |
-Half map: Half Map 2
| File | emd_40692_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
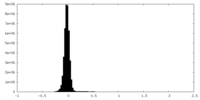
| Density Histograms |
- Sample components
Sample components
-Entire : Cleaved Ycf1p IFwide-beta Dimer
| Entire | Name: Cleaved Ycf1p IFwide-beta Dimer |
|---|---|
| Components |
|
-Supramolecule #1: Cleaved Ycf1p IFwide-beta Dimer
| Supramolecule | Name: Cleaved Ycf1p IFwide-beta Dimer / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Metal resistance protein YCF1
| Macromolecule | Name: Metal resistance protein YCF1 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO / EC number: ABC-type Cd2+ transporter |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 174.024141 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MAGNLVSWAC KLCRSPEGFG PISFYGDFTQ CFIDGVILNL SAIFMITFGI RDLVNLCKKK HSGIKYRRNW IIVSRMALVL LEIAFVSLA SLNISKEEAE NFTIVSQYAS TMLSLFVALA LHWIEYDRSV VANTVLLFYW LFETFGNFAK LINILIRHTY E GIWYSGQT ...String: MAGNLVSWAC KLCRSPEGFG PISFYGDFTQ CFIDGVILNL SAIFMITFGI RDLVNLCKKK HSGIKYRRNW IIVSRMALVL LEIAFVSLA SLNISKEEAE NFTIVSQYAS TMLSLFVALA LHWIEYDRSV VANTVLLFYW LFETFGNFAK LINILIRHTY E GIWYSGQT GFILTLFQVI TCASILLLEA LPKKPLMPHQ HIHQTLTRRK PNPYDSANIF SRITFSWMSG LMKTGYEKYL VE ADLYKLP RNFSSEELSQ KLEKNWENEL KQKSNPSLSW AICRTFGSKM LLAAFFKAIH DVLAFTQPQL LRILIKFVTD YNS ERQDDH SSLQGFENNH PQKLPIVRGF LIAFAMFLVG FTQTSVLHQY FLNVFNTGMY IKSALTALIY QKSLVLSNEA SGLS STGDI VNLMSVDVQK LQDLTQWLNL IWSGPFQIII CLYSLYKLLG NSMWVGVIIL VIMMPLNSFL MRIQKKLQKS QMKYK DERT RVISEILNNI KSLKLYAWEK PYREKLEEVR NNKELKNLTK LGCYMAVTSF QFNIVPFLVS CCTFAVFVYT EDRALT TDL VFPALTLFNL LSFPLMIIPM VLNSFIEASV SIGRLFTFFT NEELQPDSVQ RLPKVKNIGD VAINIGDDAT FLWQRKP EY KVALKNINFQ AKKGNLTCIV GKVGSGKTAL LSCMLGDLFR VKGFATVHGS VAYVSQVPWI MNGTVKENIL FGHRYDAE F YEKTIKACAL TIDLAILMDG DKTLVGEKGI SLSGGQKARL SLARAVYARA DTYLLDDPLA AVDEHVARHL IEHVLGPNG LLHTKTKVLA TNKVSALSIA DSIALLDNGE ITQQGTYDEI TKDADSPLWK LLNNYGKKNN GKSNEFGDSS ESSVRESSIP VEGELEQLQ KLNDLDFGNS DAISLRRASD ATLGSIDFGD DENIAKREHR EQGKVKWNIY LEYAKACNPK SVCVFILFIV I SMFLSVMG NVWLKHWSEV NSRYGSNPNA ARYLAIYFAL GIGSALATLI QTIVLWVFCT IHASKYLHNL MTNSVLRAPM TF FETTPIG RILNRFSNDI YKVDALLGRT FSQFFVNAVK VTFTITVICA TTWQFIFIII PLSVFYIYYQ QYYLRTSREL RRL DSITRS PIYSHFQETL GGLATVRGYS QQKRFSHINQ CRIDNNMSAF YPSINANRWL AYRLELIGSI IILGAATLSV FRLK QGTLT AGMVGLSLSY ALQITQTLNW IVRMTVEVET NIVSVERIKE YADLKSEAPL IVEGHRPPKE WPSQGDIKFN NYSTR YRPE LDLVLKHINI HIKPNEKVGI VGRTGAGKSS LTLALFRMIE ASEGNIVIDN IAINEIGLYD LRHKLSIIPQ DSQVFE GTV RENIDPINQY TDEAIWRALE LSHLKEHVLS MSNDGLDAQL TEGGGNLSVG QRQLLCLARA MLVPSKILVL DEATAAV DV ETDKVVQETI RTAFKDRTIL TIAHRLNTIM DSDRIIVLDN GKVAEFDSPG QLLSDNKSLF YSLCMEAGLV NENDYKDH D GDYKDHDIDY KDDDDK UniProtKB: Metal resistance protein YCF1 |
-Macromolecule #2: Unknown peptide
| Macromolecule | Name: Unknown peptide / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 1.124378 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) |
-Macromolecule #3: PHOSPHATIDYLETHANOLAMINE
| Macromolecule | Name: PHOSPHATIDYLETHANOLAMINE / type: ligand / ID: 3 / Number of copies: 2 / Formula: PTY |
|---|---|
| Molecular weight | Theoretical: 734.039 Da |
| Chemical component information |  ChemComp-PTY: |
-Macromolecule #4: (1R)-2-{[(S)-(2-aminoethoxy)(hydroxy)phosphoryl]oxy}-1-[(pentadec...
| Macromolecule | Name: (1R)-2-{[(S)-(2-aminoethoxy)(hydroxy)phosphoryl]oxy}-1-[(pentadecanoyloxy)methyl]ethyl (12E)-hexadeca-9,12-dienoate type: ligand / ID: 4 / Number of copies: 2 / Formula: GP7 |
|---|---|
| Molecular weight | Theoretical: 673.901 Da |
| Chemical component information | 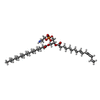 ChemComp-GP7: |
-Macromolecule #5: 2-(HEXADECANOYLOXY)-1-[(PHOSPHONOOXY)METHYL]ETHYL HEXADECANOATE
| Macromolecule | Name: 2-(HEXADECANOYLOXY)-1-[(PHOSPHONOOXY)METHYL]ETHYL HEXADECANOATE type: ligand / ID: 5 / Number of copies: 2 / Formula: LPP |
|---|---|
| Molecular weight | Theoretical: 648.891 Da |
| Chemical component information |  ChemComp-LPP: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE / Instrument: LEICA EM GP |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 46.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 4.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER Details: Cleaved Ycf1p monomer in the IFwide-beta conformation |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.2 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 71014 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)